Product Images Gabapentin
View Photos of Packaging, Labels & Appearance
- Creatinine clearance - gabapentin fig1
- Figure 1. Weekly Mean Pain Scores (Observed Cases in ITT Population): Study 1 - gabapentin fig2
- Figure 2. Weekly Mean Pain Scores (Observed Cases in ITT Population): Study 2 - gabapentin fig3
- Figure 3. Proportion of Responders (patients with ≥50% reduction in pain score) at Endpoint: Controlled PHN Studies - gabapentin fig4
- Figure 4. Responder Rate in Patients Receiving Gabapentin Expressed as a Difference from Placebo by Dose and Study: Adjunctive Therapy Studies in Patients ≥12 Years of Age with Partial Seizures - gabapentin fig5
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 600 mg (100 Tablets Bottle) - gabapentin fig6
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 800 mg (100 Tablets Bottle) - gabapentin fig7
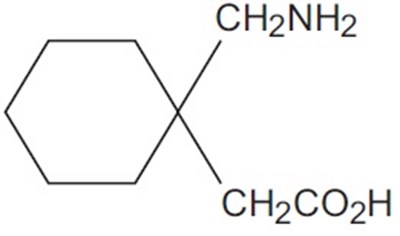
- Chemical Structure - gabapentin str
Product Label Images
The following 8 images provide visual information about the product associated with Gabapentin NDC 82009-071 by Quallent Pharmaceuticals Health Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Creatinine clearance - gabapentin fig1
This text appears to be a formula to calculate "Lt" using the age and weight of a individual, along with their creatinine level. However, without more context it is difficult to determine what "Lt" represents or what the purpose of this formula is.*
Figure 1. Weekly Mean Pain Scores (Observed Cases in ITT Population): Study 1 - gabapentin fig2

The text describes a figure depicting weekly mean pain scores observed in the ITT population for Study 1. The figure includes the names Ek, Do, Tuon, Priod, and Fred, as well as the medication Gabapentin at a dose of 300 mg/day. There is also a baseline score and a placebo treatment represented on the graph.*
Figure 3. Proportion of Responders (patients with ≥50% reduction in pain score) at Endpoint: Controlled PHN Studies - gabapentin fig4
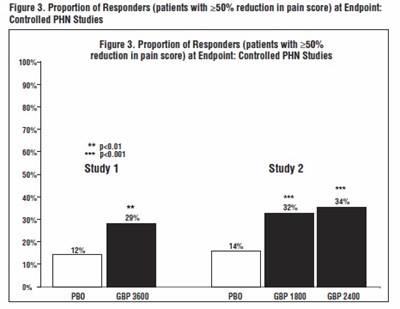
Figure 4. Responder Rate in Patients Receiving Gabapentin Expressed as a Difference from Placebo by Dose and Study: Adjunctive Therapy Studies in Patients ≥12 Years of Age with Partial Seizures - gabapentin fig5

This appears to be a table for a study on Aduncive Therapy in patients over 12 years old with partial seizures. It shows the figure expressed as a difference from placebo at various daily doses in milligrams. However, the information provided is incomplete and difficult to interpret without context.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 600 mg (100 Tablets Bottle) - gabapentin fig6
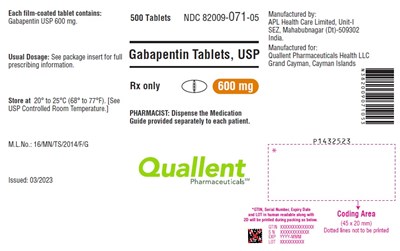
Each tablet contains 600 mg of Gabapentin USP. It is available in a package of 500 tablets. It should be stored between 20°C to 25°C. The medication guide should be provided separately for each patient. Dispensing instructions are only for pharmacists. The manufacturer is APL Health Care Limited and it is produced for Quallent Pharmaceuticals Health LLC. The expiry date of the product is not available.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 800 mg (100 Tablets Bottle) - gabapentin fig7

Gabapentin Tablets, USP containing 800 mg of Gabapentin USP. The usual dosage should be seen in the package insert for full prescribing information. The tablets should be stored at a temperature of 20°C to 25°C (66°F to 77°F) as controlled room temperature. The medication guide should be dispensed separately to each patient by a pharmacist. Manufactured by APL Health Care Limited, Unit-1 in India and distributed by Quallent Pharmaceuticals in Grand Cayman, Cayman Islands. The expiry date and lot number are not readable.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.