Product Images Adalimumab-adbm
View Photos of Packaging, Labels & Appearance
- Figure 1 - adalimumabqt 01
- Figure 2 - adalimumabqt 02
- Figure 3 - Study UV I - adalimumabqt 03
- Figure 3 - Study UV II - adalimumabqt 04
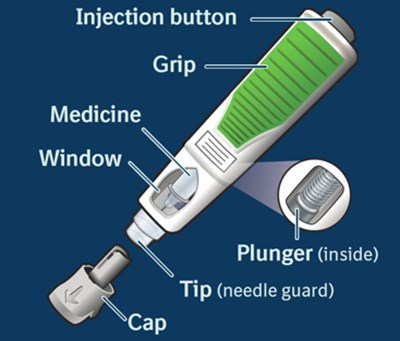
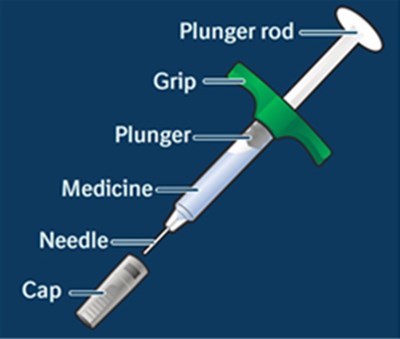
- Image - adalimumabqt 05
- Step 1 - adalimumabqt 06
- Step 2 - adalimumabqt 07
- Step 3 - adalimumabqt 08
- Step 4 - adalimumabqt 09
- Step 5 - adalimumabqt 10
- Step 6 - adalimumabqt 11
- Step 7 - adalimumabqt 12
- Step 8 - adalimumabqt 13
- Step 9 - adalimumabqt 14
- Step 10 - adalimumabqt 15
- Step 10 - adalimumabqt 16
- Step 11 - adalimumabqt 17
- Image - adalimumabqt 18
- Step 1 - adalimumabqt 19
- Step 2 - adalimumabqt 20
- Step 3 - adalimumabqt 21
- Step 4 - adalimumabqt 22
- Step 5 - adalimumabqt 23
- Step 6 - adalimumabqt 24
- Step 7 - adalimumabqt 25
- Step 8 - adalimumabqt 26
- Step 9 - adalimumabqt 27
- Step 10 - adalimumabqt 28
- Step 10 - adalimumabqt 29
- Step 11 - adalimumabqt 30
- PRINCIPAL DISPLAY PANEL - Kit Carton - NDC 82009-148-22 - adalimumabqt 31
- PRINCIPAL DISPLAY PANEL - Kit Carton - NDC 82009-144-22 - adalimumabqt 32
- PRINCIPAL DISPLAY PANEL - Kit Carton - NDC 82009-146-22 - adalimumabqt 33
- PRINCIPAL DISPLAY PANEL - Kit Carton - NDC 82009-150-22 - adalimumabqt 34
Product Label Images
The following 34 images provide visual information about the product associated with Adalimumab-adbm NDC 82009-150 by Quallent Pharmaceuticals Health Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
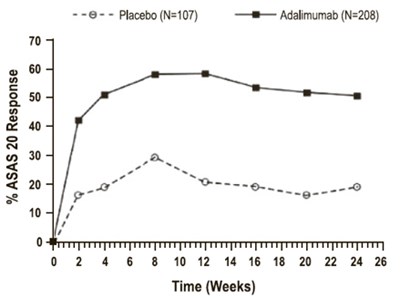
Figure 1 - adalimumabqt 01

This text provides information about the percentage of patients who are ACR 20 responders when treated with 40 mg every other week compared to those receiving a placebo.*
Step 4 - adalimumabqt 09

This text contains information about injection sites on the abdomen and thighs, helping with proper administration of injections.*
Step 9 - adalimumabqt 14

This text provides instructions for administering an injection. It guides the reader to press the injection button and hold the pen against the skin while slowly counting to 10.*
Step 7 - adalimumabqt 25

This text instructs the reader to gently press around the injection site and maintain pressure. This technique may help reduce discomfort and promote proper absorption of the injected substance.*
Step 10 - adalimumabqt 28

This text appears to be instructions related to handling a needle. It suggests removing the needle and positioning it at a 45-degree angle.*
PRINCIPAL DISPLAY PANEL - Kit Carton - NDC 82009-148-22 - adalimumabqt 31

This text appears to be a label or instructions for a medical product, likely related to a medication called Adalimumab-adbm PEN. It contains information about dosage, expiration date, and proper usage guidelines for subcutaneous administration. The product seems to come in single-dose prefilled pens and contains adalimumab. It is important to note that specific details about the medication's administration and storage are also mentioned.*
PRINCIPAL DISPLAY PANEL - Kit Carton - NDC 82009-144-22 - adalimumabqt 32

This text refers to the instructions and details of a medication called Adalimumab-adbm PEN Injection, which is intended for subcutaneous use. The medication comes in the form of two single-dose prefilled pens, each containing 40 mg of the solution. It is essential not to use this medication beyond the expiration date mentioned.*
PRINCIPAL DISPLAY PANEL - Kit Carton - NDC 82009-150-22 - adalimumabqt 34

This is a description of a medication called Adalimumab-adbm, which is an injection containing 40 mg/0.8 mL for subcutaneous use. The medication comes in 2 single-dose prefilled syringes and should not be used beyond the expiration date. It is important for each patient to receive the enclosed medication guide. The packaging includes natural rubber and dry syringes. The text provides detailed instructions and numbers related to dosage and administration.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.