Product Images Catapres-tts-2
View Photos of Packaging, Labels & Appearance
- PRINCIPAL DISPLAY PANEL - 0.1 mg Adhesive Cover Pouch - catapres 01a
- PRINCIPAL DISPLAY PANEL - 0.2 mg Adhesive Cover Pouch - catapres 03a
- PRINCIPAL DISPLAY PANEL - 0.3 mg Adhesive Cover Pouch - catapres 05a
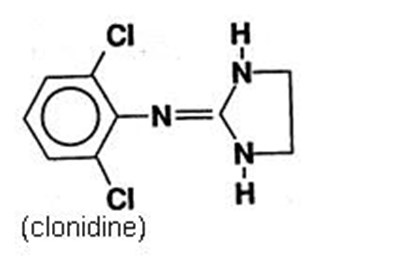
- catapres-tts-structure - catapres tts structure
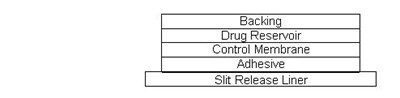
- cross-section-table - cross section table
- figure-1 - figure 1
- figure-2 - figure 2
- figure-3 - figure 3
- figure-4 - figure 4
- figure-5 - figure 5
- figure-6 - figure 6
- PRINCIPAL DISPLAY PANEL - 0.1 mg Patch Pouch Label - tn catapres 01 ptch
- PRINCIPAL DISPLAY PANEL - 0.2 mg Patch Pouch Label - tn catapres 02 ptch
- PRINCIPAL DISPLAY PANEL - 0.3 mg Patch Pouch Label - tn catapres 03 ptch
- PRINCIPAL DISPLAY PANEL - 0.1 mg Patch Pouch Carton - tncatapres 01
- PRINCIPAL DISPLAY PANEL - 0.2 mg Patch Pouch Carton - tncatapres 02 bx
- PRINCIPAL DISPLAY PANEL - 0.3 mg Patch Pouch Carton - tncatapres 03 bx
Product Label Images
The following 17 images provide visual information about the product associated with Catapres-tts-2 NDC 82089-102 by Technomed Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
PRINCIPAL DISPLAY PANEL - 0.1 mg Adhesive Cover Pouch - catapres 01a

This is a warning about an adhesive cover that does not contain active medication. The cover should be applied over the patch if necessary. There are patient instructions available for use of the adhesive cover. The product code is L15000B 10003925/02.*
PRINCIPAL DISPLAY PANEL - 0.2 mg Adhesive Cover Pouch - catapres 03a
This appears to be a product label or packaging insert for an adhesive cover containing no active medication, to be used as per patient instructions. The label contains several warnings, instructions and a product code.*
PRINCIPAL DISPLAY PANEL - 0.3 mg Adhesive Cover Pouch - catapres 05a

This is a warning label for an adhesive cover that should be used for external application only. It does not contain any active medication. It is advised to refer to the patient instructions before use. The product code is L15002A 10003923/02.*
figure-2 - figure 2

This appears to be a medication packaging label that includes a patch. The label suggests that the package contains a patch with medication and also includes a cover that can be used if the patch becomes loose.*
PRINCIPAL DISPLAY PANEL - 0.1 mg Patch Pouch Label - tn catapres 01 ptch

Catapres-TTS is a transdermal system that delivers clonidine in vivo to provide 0.1mg per day for seven days consecutively. The transdermal system should be removed before undergoing an MRI procedure. Each patch contains one medication, and you should remove the clear backing before using it. The patch should be stored at controlled room temperature, between 15°C to 30°C (59°F to 86°F). The manufacturer of Catapres-TTS is Alza Corporation, Vacaville, CA.*
PRINCIPAL DISPLAY PANEL - 0.2 mg Patch Pouch Label - tn catapres 02 ptch

Catapres-TTS"-2 is a transdermal system that delivers 0.2mg clonidine per day for one week. The patch should be removed before an MRI procedure to avoid burns. The medication is contained in a single patch that should be stored within the temperature range of 15°C-30°C. The product is manufactured by Alza Corporation, and Catapres-TTS is a registered trademark of Boehringer Ingelheim International GmbH.*
PRINCIPAL DISPLAY PANEL - 0.3 mg Patch Pouch Label - tn catapres 03 ptch

Catapres-TTS®-3 is a transdermal system that delivers clonidine in vivo at a rate of 0.3 mg per day for one week. The patch needs to be removed before undergoing an MRI procedure. The pouch contains one patch with medication. The clear backing needs to be removed before using it. It needs to be stored at 25°C (77°F) with permitted excursions to 15°-30°C (59°-86°F). The product is manufactured by Alza Corporation in Vacaville, CA 95688, USA. Tear here to remove the patch.*
PRINCIPAL DISPLAY PANEL - 0.1 mg Patch Pouch Carton - tncatapres 01

This text contains a mix of random errors and incomplete sentences. As a result, it does not convey any useful information and cannot be evaluated.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.