FDA Label for Pain Relieving
View Indications, Usage & Precautions
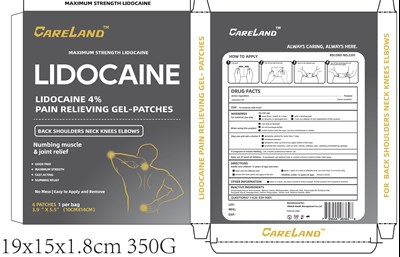
Pain Relieving Product Label
The following document was submitted to the FDA by the labeler of this product Uniaid Health Management (suzhou) Co.,ltd. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Drug Facts
Active Ingredient
Lidocaine 4.0%
Purpose
Topical anesthetic
Use
For temporary relief of pain
Warnings
For external use only
Do Not Use
- more than 1 patch at a time
- with a heating pad
- on wounds or damaged skin
- if you are allergic to any ingredients of this product
When Using This Product
- use only as directed
- do not bandage tightly
- avoid contact with the eyes, mucous membranes or rashes
Stop Use And Ask A Doctor If
- symptoms persist for more than 7 days
- conditions worsen
- symptoms clear up and occur again within a few days
- localized skin reactions, such as rash, itching, redness, pain, swelling and blistering develop
If Pregnant Or Breast Feeding,
ask a health professional before use
Keep Out Of Reach Of Children.
If swallowed, get medical help or contact a Poison Control Center right away.
Directions
Adults and children 12 years of age and over:
- clean and dry affected area
- apply 1 patch at a time to affected area, not more than 3 to 4 times daily
- remove film from patch and apply to the skin
Children under 12 years of age: consult a doctor
Other Information
- Store in a clean, dry place outside of direct sunlight, Protect product from excessive moisture
Inactive Ingredient
Dihydroxyaluminum Aminoacetate, Glycerol, Kaolin, Methylparaben, Polyacrylic Acid, Polysorbate 80, Povidone K90, Propylene Glycol, Propylparaben, Sodium Polyacrylate, Tartaric Acid, Titanium Dioxide, Water
Questions?
1-626-839-9001
Package Labeling:
* Please review the disclaimer below.