Product Images Betapace
View Photos of Packaging, Labels & Appearance
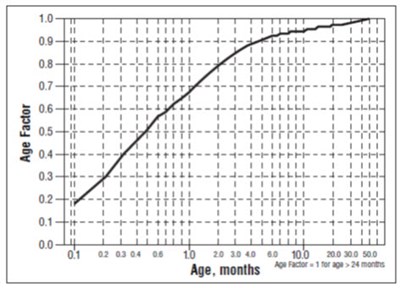
- Figure 1 - image 01
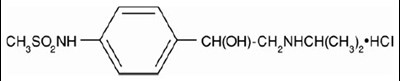
- Structural Formula - image 02
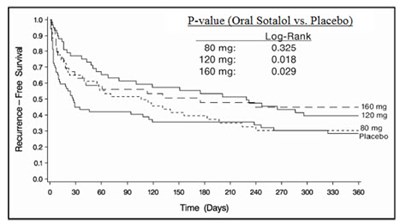
- Figure 2 - image 03
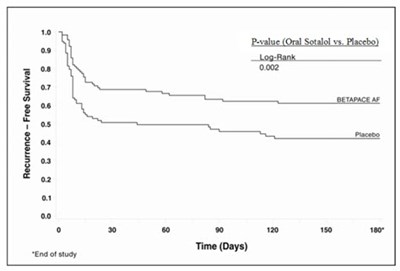
- Figure 3 - image 04
- Principal Display Panel - 80 mg Betapace - image 06
- Principal Display Panel - 120 mg Betapace - image 07
- Principal Display Panel - 160 mg Betapace - image 08
- Principal Display Panel - 80 mg Betapace AF - image 09
- Principal Display Panel - 120 mg Betapace AF - image 10
- Principal Display Panel - 160 mg Betapace AF - image 11
Product Label Images
The following 10 images provide visual information about the product associated with Betapace NDC 83107-005 by Legacy Pharma Usa, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Principal Display Panel - 80 mg Betapace - image 06
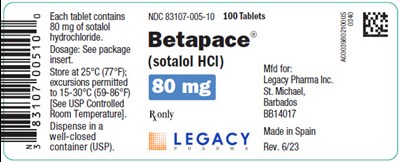
Each tablet contains 80 mg of sotalol hydrochloride. The recommended dosage can be found in the package insert. Store the tablets at a temperature of 25°C (77°F), with excursions permissible between 15-30°C (50-86°F) according to USP Controlled Room Temperature guidelines. Dispense the tablets in a well-closed container as per USP regulations. The product is identified with NDC 83107-005-10 and contains 100 tablets. The brand name of the medication is Betapace® (sotalol HCI). It is manufactured by Legacy Pharma Inc. located in St. Michael, Barbados. The product is made in Spain.*
Principal Display Panel - 120 mg Betapace - image 07

Each tablet contains 120 mg of sotalol hydrochloride. Dosage information can be found in the package insert. The tablets should be stored at a temperature of 25°C (77°F) with excursions permitted between 15-30°C (50-86°F) according to USP Controlled Room Temperature guidelines. They should be dispensed in a well-closed container. The product is identified with NDC 83107-006-10 and comes in a package of 100 tablets. The brand name of the product is Betapace® (sotalol HCI). It is manufactured for Legacy Pharma Inc. and made in Spain.*
Principal Display Panel - 160 mg Betapace - image 08

Each tablet contains 160 mg of sotalol hydrochloride. This medication, known as Betapace, is manufactured by Ronly LEGACY and dispensed in a well-closed container. The recommended dosage can be found in the package insert. It should be stored at a temperature of 25°C (77°F), with excursions allowed between 15-30°C (59-86°F) within USP Controlled Room Temperature guidelines. The product is labeled with NDC 83107-007-10 and is made in Spain. The manufacturer is Legacy Pharma nc, located in St. Michael, Barbados.*
Principal Display Panel - 80 mg Betapace AF - image 09

Each tablet contains 80 mg of sotalol hydrochloride. This medication is used to treat certain types of irregular heartbeat. The dosage should be taken as prescribed by your physician. For more information, please refer to the package insert. The tablets should be stored at a temperature of 25°C (77°F), with permitted excursions to 15-30°C (59-86°F). The product is packaged in a unit of 60 tablets. It is manufactured by Legacy Pharma Inc. in St. Michael, Barbados, and made in Spain.*
Principal Display Panel - 160 mg Betapace AF - image 11

Each tablet contains 160 mg of sotalol hydrochloride. This medication should be taken as prescribed by your physician. Please refer to the package insert for more information. The tablets should be stored at a temperature of 25°C (77°F), with excursions permitted between 15-30°C (59-86°F). The NDC code for the medication is 83107-010-60 and it is available in a unit pack of 60 tablets. The manufacturer is Legacy Pharma Inc., located in St. Michael, Barbados, and the medication is made in Spain. Please consult your healthcare provider or pharmacist for further details.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.