Product Images Ogivri
View Photos of Packaging, Labels & Appearance
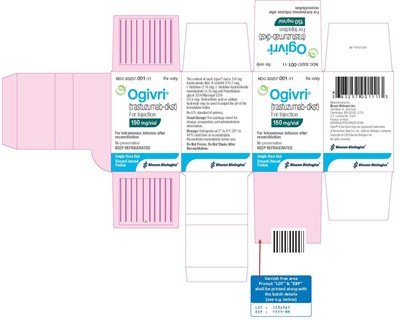
- Figure 10 Ogivri for Injection 150 mg/vial Carton Label - Image 10
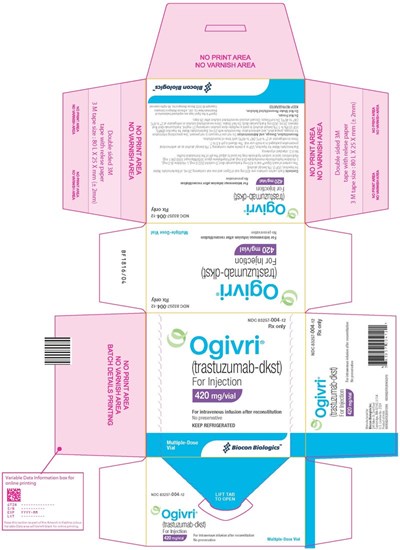
- Figure 8 Ogivri for Injection 420 mgvial Carton Label with diluent - Image 8
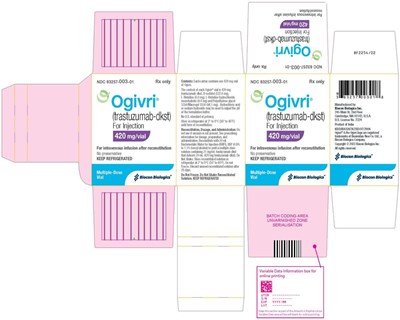
- Figure 9 Ogivri for Injection 420 mg/vial Carton Label - Image 9
- Figure 1 Studies 1 and 2: Cumulative Incidence of Time to First LVEF - image 01
- Figure 2 Study 3: Cumulative Incidence of Time to First LVEF Decline of ≥ 10 Percentage Points from Baseline and to Below 50% with Death as a Competing Risk Event - image 02
- Figure 3 Study 4: Cumulative Incidence of Time to First LVEF Decline of ≥ 10 Percentage Points from Baseline and to Below 50% with Death as a Competing Risk Event - image 03
- Figure 4 Duration of Disease-Free Survival in Patients with Adjuvant Treatment of Breast Cancer (Studies 1 and 2) - image 04
- Figure 5 Duration of Overall Survival in Patients with Adjuvant Treatment of Breast Cancer (Studies 1 and 2) - image 05
- Figure 6 Duration of Disease-Free Survival in Patients with Adjuvant Treatment of Breast Cancer (Study 4) - image 06
- Figure 7 Updated Overall Survival in Patients with Metastatic Gastric Cancer (Study 7) - image 07
Product Label Images
The following 10 images provide visual information about the product associated with Ogivri NDC 83257-004 by Biocon Biologics Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 2 Study 3: Cumulative Incidence of Time to First LVEF Decline of ≥ 10 Percentage Points from Baseline and to Below 50% with Death as a Competing Risk Event - image 02

Figure 3 Study 4: Cumulative Incidence of Time to First LVEF Decline of ≥ 10 Percentage Points from Baseline and to Below 50% with Death as a Competing Risk Event - image 03

Figure 4 Duration of Disease-Free Survival in Patients with Adjuvant Treatment of Breast Cancer (Studies 1 and 2) - image 04

Figure 5 Duration of Overall Survival in Patients with Adjuvant Treatment of Breast Cancer (Studies 1 and 2) - image 05

Figure 6 Duration of Disease-Free Survival in Patients with Adjuvant Treatment of Breast Cancer (Study 4) - image 06

Figure 6 shows the duration of disease-free survival in patients with adjuvant treatment of breast cancer in Study 4. The graph displays different treatment combinations, including AC->T (doxorubicin + cyclophosphamide followed by docetaxel and trastuzumab), AC->TH (doxorubicin + cyclophosphamide followed by docetaxel, platinum salt, and trastuzumab), and TCH (docetaxel, platinum salt, and trastuzumab). The x-axis represents the number of years of disease-free survival, while the y-axis represents the number of patients. The Kaplan-Meier estimates are used to calculate the survival rates.*
Figure 7 Updated Overall Survival in Patients with Metastatic Gastric Cancer (Study 7) - image 07

Not available.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.