Product Images Humira
View Photos of Packaging, Labels & Appearance
- Figure 1. Study RA-III ACR 20 Responses over 52 Weeks - humira cordavis co branded 01
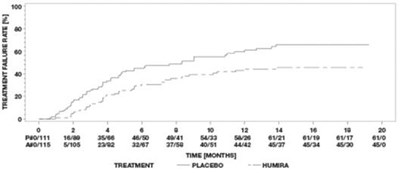
- Figure 2. ASAS 20 Response By Visit, Study AS-I - humira cordavis co branded 02
- Title: km-graph-fig3-study-uv-i - humira cordavis co branded 03
- Title: uveitis-km-fig3-part2 - humira cordavis co branded 04
- Title: km-peds-uv - humira cordavis co branded 05
- Title: fig-a-ifu-40mg-pen - humira cordavis co branded 06
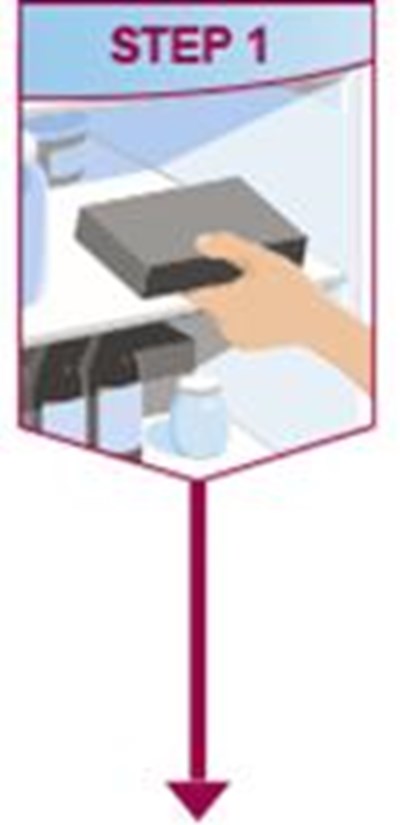
- Title: step1-pen-ifu-40mg - humira cordavis co branded 07
- Title: ifu-40mg-0-4ml-pen-step2 - humira cordavis co branded 08
- Title: humira mai ifu resize step3 - humira cordavis co branded 09
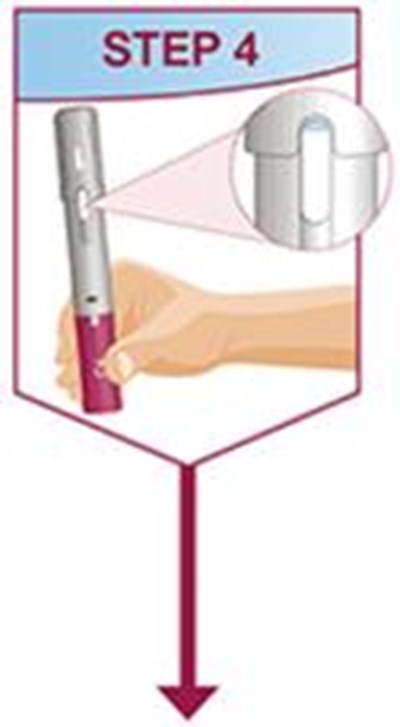
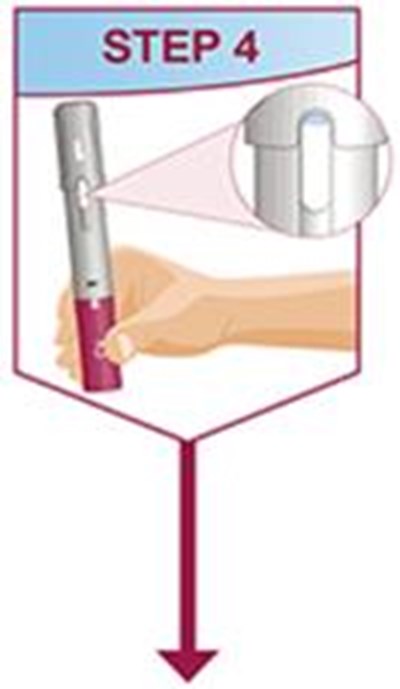
- Title: humira mai ifu resize1 step4 - humira cordavis co branded 0a
- Title: humira-mai-ifu-resize1-step5 - humira cordavis co branded 0b
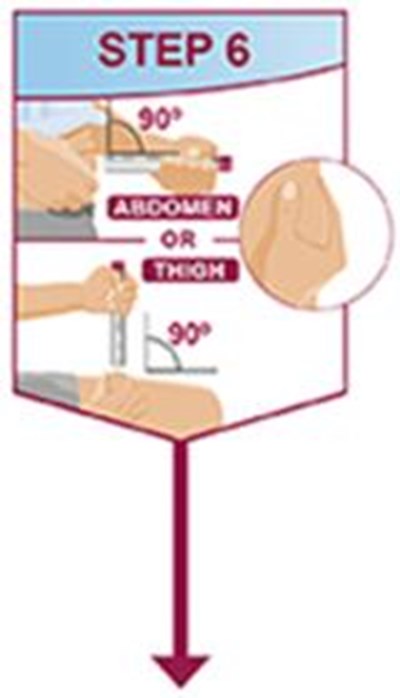
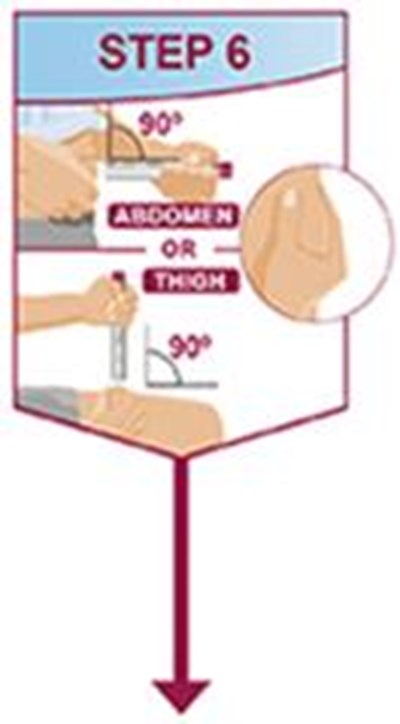
- Title: ifu-40mg-0-4ml-pen-step6 - humira cordavis co branded 0c
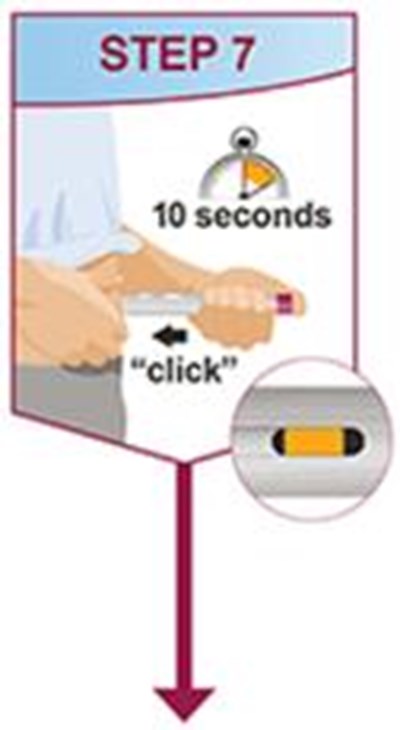
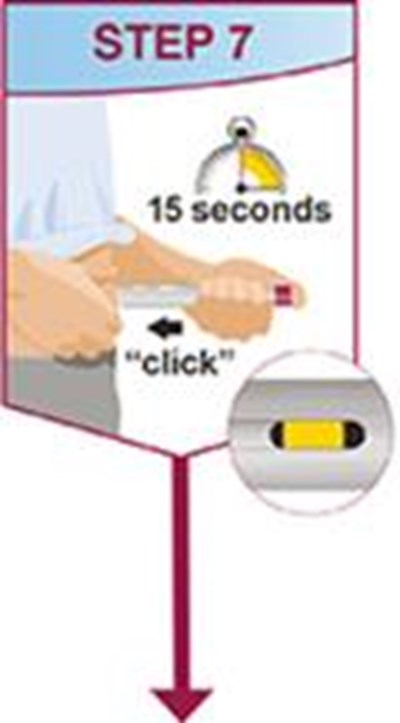
- Title: humira mai ifu resize1 step7 - humira cordavis co branded 0d
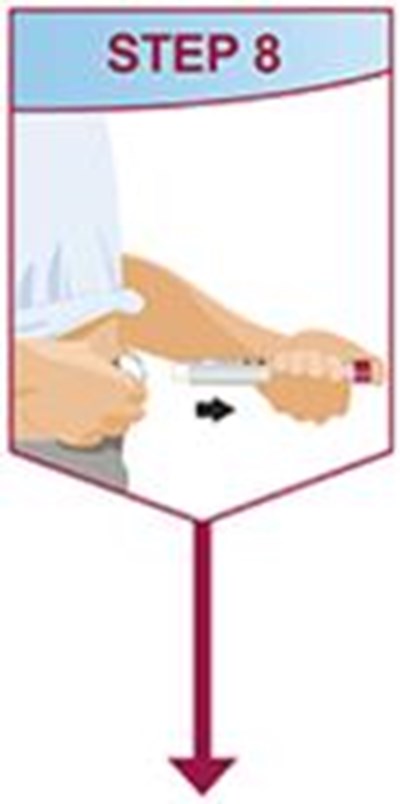
- Title: humira mai ifu resize1 step8 - humira cordavis co branded 0e
- Title: ifu-40mg0-4ml-pen-step9 - humira cordavis co branded 0f
- Title: humira mai ifu resize1 calendar - humira cordavis co branded 10
- Title: humira mai ifu abbvie logo - humira cordavis co branded 11
- Title: step1-80mg-ifu - humira cordavis co branded 12
- Title: 80mg-mai-ifu-step4 - humira cordavis co branded 13
- Title: ifu-80mg-0-8ml-pen-step6 - humira cordavis co branded 14
- Title: 80mg-mai-ifu-step7-arrow - humira cordavis co branded 15
- Title: 80mg-mai-ifu-logo - humira cordavis co branded 16
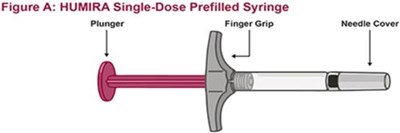
- Title: fig-a-exploded-pfs - humira cordavis co branded 17
- Title: ifu-single-dpse-pfs-step2 - humira cordavis co branded 18
- Title: syringe-pms-step3 - humira cordavis co branded 19
- Title: step4-pfs-ifu - humira cordavis co branded 1a
- Title: syringe-pms-step5 - humira cordavis co branded 1b
- Title: syringe-pms-step6 - humira cordavis co branded 1c
- Title: new-step-7-syringes - humira cordavis co branded 1d
- Title: step8-ifu-pfs - humira cordavis co branded 1e
- Title: ifu-single-dose-pfs-step9 - humira cordavis co branded 1f
- Title: syringe-ifu-pms-abbvie-logo - humira cordavis co branded 20
- A close-up of a packageDescription automatically generated - humira cordavis co branded 21
- A close-up of a prescription medicineDescription automatically generated - humira cordavis co branded 22
- A label of a medicineDescription automatically generated with medium confidence - humira cordavis co branded 23
- A close-up of a labelDescription automatically generated - humira cordavis co branded 24
- A label of a medicineDescription automatically generated - humira cordavis co branded 25
Product Label Images
The following 37 images provide visual information about the product associated with Humira NDC 83457-554 by Cordavis Limited, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1. Study RA-III ACR 20 Responses over 52 Weeks - humira cordavis co branded 01
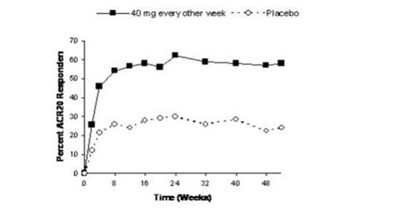
The text is not available.*
Figure 2. ASAS 20 Response By Visit, Study AS-I - humira cordavis co branded 02
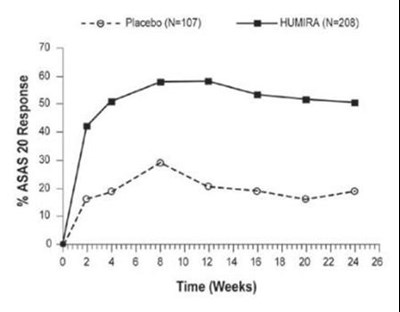
This is a comparison chart showing the response rate to the ASAS 20 criteria between a placebo group (N=107) and a group treated with HUMIRA (N=208) over a span of 24 weeks. The response rate is observed at different time points.*
Title: fig-a-ifu-40mg-pen - humira cordavis co branded 06

This is a description of the HUMIRA Single-Dose Pen. It includes details such as the gray cap, white needle sleeve, plum-colored cap, inspection window for the needle, and an activator button.*
A close-up of a packageDescription automatically generated - humira cordavis co branded 21

This is a description of a medical product called HUMIRA PEN, which is a subcutaneous medication containing adalimumab. It comes in a carton with 2 single-dose prefilled pens for use by patients. The medication is intended for subcutaneous use only and each pen contains 1 ml solution. Pharmacists are required to provide patients with an enclosed Medication Guide. To open the package, instructions from abbvie can be followed.*
A close-up of a prescription medicineDescription automatically generated - humira cordavis co branded 22
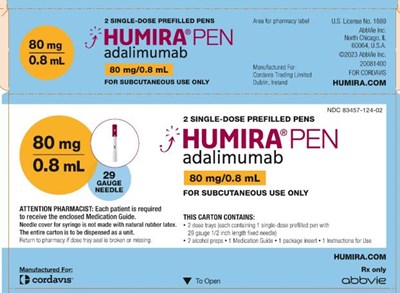
This is information about a medication called "HUMIRA PEN" containing adalimumab for subcutaneous use. The medication comes in a package that includes 2 single-dose prefilled pens. The text also requests pharmacists to provide the included Medication Guide to each patient receiving the medication. It mentions that the pen does not contain natural rubber latex and provides details about the pen's components. The packaging information implies that each pen should be used as directed.*
A label of a medicineDescription automatically generated with medium confidence - humira cordavis co branded 23

This text appears to be a medication label for Humira (adalimumab) containing information about the dosage and administration for subcutaneous use. It includes details about the content, such as the prefilled syringes with a specific dose of 10 mg/0.4 mL of adalimumab and instructions for use. It also mentions the requirement for patients to receive a Medication Guide and notes on the packaging materials.*
A close-up of a labelDescription automatically generated - humira cordavis co branded 24

This is a description of a medication called HUMIRA® in a prefilled syringe form with 20 mg/0.2 mL of adalimumab for subcutaneous use. Each package contains 2 single-dose prefilled syringes and a Medication Guide. The syringe cover is not made with natural rubber latex. Patients are instructed to visit HUMIRA.COM for more information. The medication is for prescription use only.*
A label of a medicineDescription automatically generated - humira cordavis co branded 25

This description provides information about a medication called HUMIRA, which comes in the form of two single-dose prefilled syringes containing adalimumab. The dosage strength is 40 mg/0.4 mL. It is intended for subcutaneous use only. The medication is manufactured for Rx only and the enclosed Medication Guide should be given to each patient. The syringes are not made with natural rubber latex. The information also includes the manufacturer's details and instructions to visit HUMIRA.COM for more information.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.