FDA Label for Adi Medical Pvp-i Pouch, Foil
View Indications, Usage & Precautions
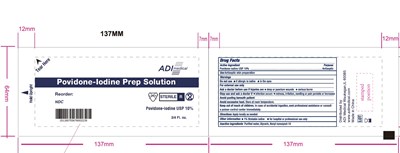
Adi Medical Pvp-i Pouch, Foil Product Label
The following document was submitted to the FDA by the labeler of this product Asia Dynamics Inc., D/b/a Adi Medical. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Active Ingredient
Povidone-lodine USP 10%
Purpose
Antiseptic
Use
for antiseptic skin preparation
Warnings
For external use only
Avoid pooling beneath patient
Do Not Use
- if allergic to lodine
- in the eye
Stop Use And Ask A Doctor If
- redness, irritation, swelling or pain persists or increases
- infection occurs
Keep Out Of Reach Of Children.
In case of accidental ingestion, seek professional assistance or consult a poison control center immediately.
Ask A Doctor Before Use If Injuries Are
- deep or puncture wounds
- serious burns
Directions
apply locally as needed
Other Information
- 1% titratable iodine
- for hospital or professional use only
Inactive Ingredients:
Nonyl Nonoxynol-10, Glycerin, Purifed Water.
Storage
Avoid excessive heat
Store at room temperature
Package Labeling:
NDC 83645-552-11
ADI Medical
* Please review the disclaimer below.