FDA Label for Hyqvia
View Indications, Usage & Precautions
- PRINCIPAL DISPLAY PANEL - 2.5 G/25 ML VIAL LABEL
- PRINCIPAL DISPLAY PANEL - 1.25 ML VIAL LABEL
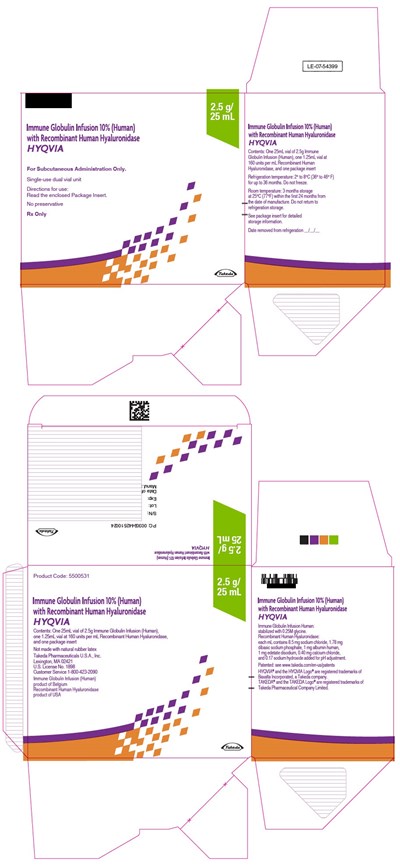
- PRINCIPAL DISPLAY PANEL - 2.5 G/25 ML KIT CARTON
- PRINCIPAL DISPLAY PANEL - 5 G/50 ML VIAL LABEL
- PRINCIPAL DISPLAY PANEL - 2.5 ML VIAL LABEL
- PRINCIPAL DISPLAY PANEL - 5 G/50 ML KIT CARTON
- PRINCIPAL DISPLAY PANEL - 10 G/100 ML VIAL LABEL
- PRINCIPAL DISPLAY PANEL - 5 ML VIAL LABEL
- PRINCIPAL DISPLAY PANEL - 10 G/100 ML KIT CARTON
- PRINCIPAL DISPLAY PANEL - 10 ML VIAL LABEL
- PRINCIPAL DISPLAY PANEL - 15 ML VIAL LABEL
Hyqvia Product Label
The following document was submitted to the FDA by the labeler of this product Bamboo Us Bidco Llc. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Principal Display Panel - 2.5 G/25 Ml Vial Label
Immune Globulin Infusion 10% (Human)
Component of HYQVIA
Single-Use Vial
Rx Only
Takeda Pharmaceuticals U.S.A., Inc.
Lexington, MA 02421
U.S. License No. 1898
Infuse 2nd
2.5 g/
25 mL
IG
LE-07-54397
Takeda
Principal Display Panel - 1.25 Ml Vial Label
Recombinant Human Hyaluronidase
Component of HYQVIA
Single-Use Vial
Rx Only
Takeda Pharmaceuticals U.S.A., Inc.
Lexington, MA 02421
Infuse 1st
HY
1.25 mL
160 units per mL
Takeda
Principal Display Panel - 2.5 G/25 Ml Kit Carton
Immune Globulin Infusion 10% (Human)
with Recombinant Human Hyaluronidase
HYQVIA
For Subcutaneous Administration Only.
Single-use dual vial unit
Directions for use:
Read the enclosed Package Insert.
No preservative
Rx Only
2.5 g/
25 mL
Takeda
Principal Display Panel - 5 G/50 Ml Vial Label
Immune Globulin Infusion 10% (Human)
Component of HYQVIA
Single-Use Vial
Rx Only
Takeda Pharmaceuticals U.S.A., Inc.
Lexington, MA 02421
U.S. License No. 1898
Infuse 2nd
5 g/
50 mL
IG
LE-07-54409
Takeda
Principal Display Panel - 2.5 Ml Vial Label
Recombinant Human Hyaluronidase
Component of HYQVIA
Single-Use Vial
Rx Only
Takeda Pharmaceuticals U.S.A., Inc.
Lexington, MA 02421
Infuse 1st
HY
2.5 mL
160 units per mL
Takeda
Principal Display Panel - 5 G/50 Ml Kit Carton
Immune Globulin Infusion 10% (Human)
with Recombinant Human Hyaluronidase
HYQVIA
For Subcutaneous Administration Only.
Single-use dual vial unit
Directions for use:
Read the enclosed Package Insert.
No preservative
Rx Only
5 g/
50 mL
Takeda
Principal Display Panel - 10 G/100 Ml Vial Label
Immune Globulin Infusion 10% (Human)
Component of HYQVIA
Single-Use Vial
Rx Only
Takeda Pharmaceuticals U.S.A., Inc.
Lexington, MA 02421
U.S. License No. 1898
Infuse 2nd
10 g/
100 mL
IG
LE-07-54412
Takeda
Principal Display Panel - 5 Ml Vial Label
Recombinant Human Hyaluronidase
Component of HYQVIA
Single-Use Vial
Rx Only
Takeda Pharmaceuticals U.S.A., Inc.
Lexington, MA 02421
Infuse 1st
5 mL
160 units per mL
HY
Takeda
Principal Display Panel - 10 G/100 Ml Kit Carton
Immune Globulin Infusion 10% (Human)
with Recombinant Human Hyaluronidase
HYQVIA
For Subcutaneous Administration Only.
Single-use dual vial unit
Directions for use:
Read the enclosed Package Insert.
No preservative
Rx Only
10 g/
100 mL
Takeda
Principal Display Panel - 10 Ml Vial Label
Recombinant Human Hyaluronidase
Component of HYQVIA
Single-Use Vial
Rx Only
Takeda Pharmaceuticals U.S.A., Inc.
Lexington, MA 02421
Infuse 1st
HY
10 mL
160 units per mL
LE-07-54417
Takeda
Principal Display Panel - 15 Ml Vial Label
Recombinant Human Hyaluronidase
Component of HYQVIA
Single-Use Vial
Rx Only
Takeda Pharmaceuticals U.S.A., Inc.
Lexington, MA 02421
Infuse 1st
HY
15 mL
160 units per mL
LE-07-54420
Takeda
* Please review the disclaimer below.