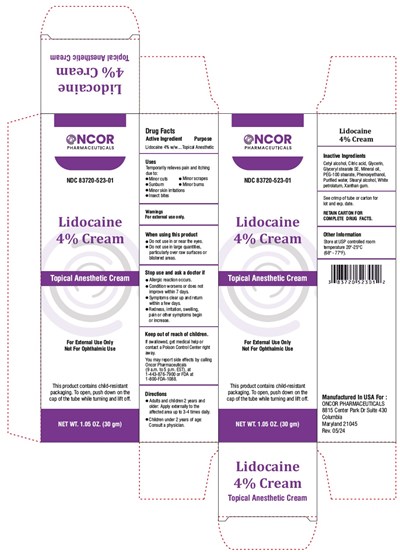
FDA Label for Lidocaine 4% Topical Anesthetic Cream
View Indications, Usage & Precautions
Lidocaine 4% Topical Anesthetic Cream Product Label
The following document was submitted to the FDA by the labeler of this product Oncor Pharmaceuticals. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Drug Facts Active Ingredients
Lidocaine 4% w/w
Purpose
Topical Anesthetic
Uses
Temporarily relieves pain and itching due to:
- Minor cuts
- Minor scrapes
- Sunburn
- Minor burns
- Minor skin irritations
- Insect bites
Warnings
For external use only.
When Using This Product
- Do not use in or near the eyes.
- Do not use in large quantities, particularly over raw surfaces or blistered areas.
Stop Use And Ask A Doctor If
- Allergic reaction occurs.
- Condition worsens or does not improve within 7 days.
- Symptoms clear up and return within a few days.
- Redness, irritation, swelling, pain or other symptoms begin or increase.
Keep Out Of Reach Of Children
If swallowed, get medical help or contact a Poison Control Center right away.
You may report side effects by calling Oncor Pharmaceuticals (9 a.m. to 5 p.m. EST), at 1-443-876-7900 or FDA at 1-800-FDA-1088.
Directions
- Adults and children 2 years and older: apply externally to the affected area up to 3-4 times daily.
- Children under 2 years of age: consult a physician.
Other
Lidocaine
4% Cream
See crimp of tube or carton for lot and exp. date.
RETAIN CARTON FOR COMPLETE DRUG FACTS.
Inactive Ingredients
Cetyl alcohol, Citric acid, Glycerin, Glyceryl stearate SE, Mineral oil, PEG-100 stearate, Phenoxyethanol, Purified water, Stearyl alcohol, White
petrolatum, Xanthan gum.
Other Information
Store at USP controlled room temperature 20°-25°C (68°-77°F).
Manufactured In USA For :
ONCOR PHARMACEUTICALS
8815 Center Park Dr Suite 430
Columbia
Maryland 21045
Rev. 05/24
Package Label
ONCOR PHARMACEUTICALS
NDC 83720- 523-01
Lidocaine 4% Cream
Topical Anesthetic Cream
For External Use Only
Not For Ophthalmic Use
This product contains child-resistant
packaging. To open, push down on the cap of the tube while turning and lift off.
NET WT. 1.05 OZ. (30 gm)
ONCOR PHARMACEUTICALS
NDC 83720- 523-01
Lidocaine 4% Cream
Topical Anesthetic Cream
For External Use Only
Not For Ophthalmic Use
This product contains child-resistant
packaging. To open, push down on the cap of the tube while turning and lift off.
NET WT. 1.05 OZ. (30 gm)
* Please review the disclaimer below.