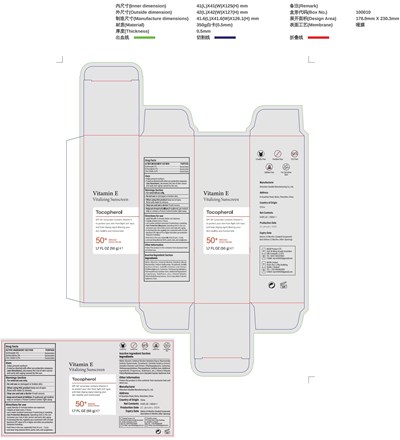
FDA Label for Vitamin E Vitalizing Sunscreen Tocopherol
View Indications, Usage & Precautions
Vitamin E Vitalizing Sunscreen Tocopherol Product Label
The following document was submitted to the FDA by the labeler of this product Shenzhen Xiaomai Manufacturing Co., Ltd.. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Active Ingredient
Niacinamide: 2%
Sodium Hyaluronate: 0.5%
Hyaluronic Acid: 0.1%
Tocopherol: 0.2%
Ethylhexyl Methoxycinnamate: 5%
Octocrylene: 4%
Zinc Oxide: 3%
Centella Asiatica Extract: 1%
Camellia Sinensis Leaf Extract: 1%
Purpose
helps prevent sunburn.
Uses
decreases the risk of skin cancer and early skin aging caused by the sun.
Warnings
For external use only.
Do Not Use
on damaged or broken skin.
When Using This Product
keep out of eyes.
Rinse with water to remove.
Stop Use And Ask A Doctor
if rash occurs.
Keep Out Of Reach Of Children.
If swallowed, get medicalhelp or contact a Poison Control Center right away.
Directions For Use
apply liberally 15 minutes before sun exposure.
reapply at least every 2 hours.
use a water resistant sunscreen if swimming or sweating.
Inactive Ingredient
Aqua: 65%
Glycerin: 3%
Cetearyl Alcohol: 2%
Butylene Glycol: 3%
Ethylhexylglycerin: 0.1%
Xanthan Gum: 0.2%
Phenoxyethanol: 0.8%
Carbomer: 0.2%
OTV: 5%
Triethoxycaprylylsilane: 0.1%
Dibutyl Adipate: 2%
Polymethylsilsesquioxane: 1.7%
Linalool: 0.1%
Other Information
Protect the product in this container from excessive heat and direct sun.
Package Label.Principal Display Panel
* Please review the disclaimer below.