FDA Label for Eye Wash
View Indications, Usage & Precautions
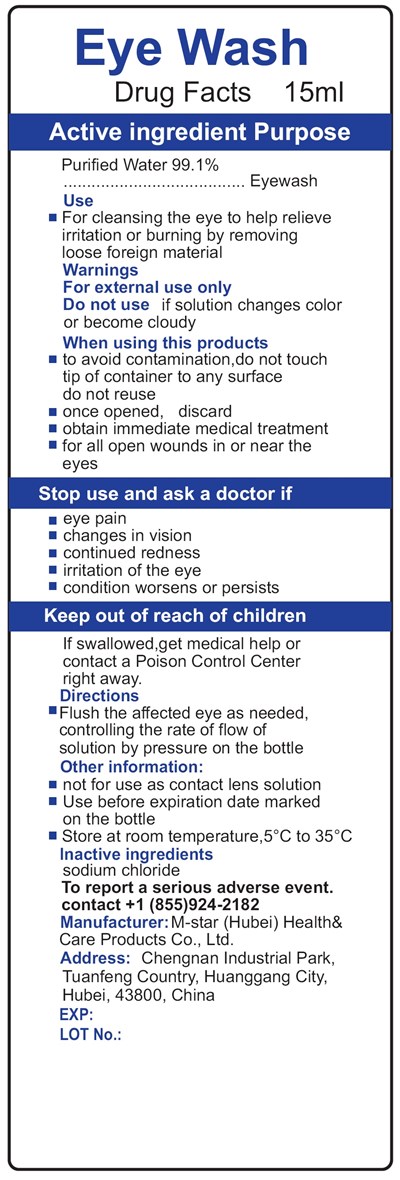
Eye Wash Product Label
The following document was submitted to the FDA by the labeler of this product M-star (hubei) Health&care Products Co., Ltd.. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Drug Facts
Active Ingredient
Purified Water 99.1%
Purpose
Eyewash
Use
- For cleansing the eye to help relieve irritation or burning by removing loose foreign material
Warnings
For external use only.
Do Not Use
if solution changes color or become cloudy
When Using This Products
- to avoid contamination,do not touch tip of container to any surface do not reuse
- once opened, discard
- obtain immediate medical treatment
- for all open wounds in or near the eyes
Stop Use And Ask A Doctor If
- eye pain
- changes in vision
- continued redness
- irritation of the eye
- condition worsens or persists
Keep Out Of Reach Of Children.
If swallowed, get medical help or contact a Poison Control Center right away.
Directions
- Flush the affected eye as needed, controlling the rate of flow of solution by pressure on the bottle
Other Information
- not for use as contact lens solution
- Use before expiration date marked on the bottle
- Store at room temperature,5°C to 35°C
Inactive Ingredients
sodium chloride
To Report A Serious Adverse Event.
contact +1(855)924-2182
Package Labelling:
* Please review the disclaimer below.