Product Images Diclofenac Sodium 1.5%
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 8 images provide visual information about the product associated with Diclofenac Sodium 1.5% NDC 85509-1038 by Phoenix Rx Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
label - Diclofenac1.5 sola 1038

This is a description of a topical solution containing diclofenac sodium USP. Each mL of the solution contains 16.05 mg of diclofenac sodium along with various excipients. The product is intended for external use only. It comes in a 5 fl. oz. (150mL) container and should be dispensed with a Medication Guide to each patient. It is manufactured by SOLA Pharmaceuticals, LLC and repackaged by PHOENIX RX LLC in Hatboro, PA. The solution is labeled with NDC# 8550910381 and includes additional information for dosage, administration, and warnings related to skin irritation.*
EFF 2 - Efficacy 2 copy
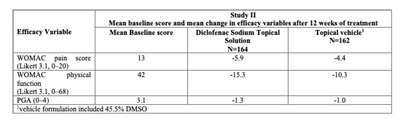
This text appears to be related to a study or evaluation involving the WOMAC pain score, WOMAC physical function, and PGA (Patient Global Assessment). It also mentions a psychosocial formulation and the inclusion of a certain percentage of DMSO. The context of the study or evaluation seems to be focused on assessing efficacy variables.*
EFFICACY - Efficacy Variable copy
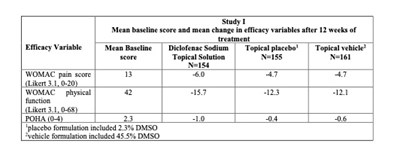
The text provides information on a study that evaluated the efficacy of diclofenac sodium and topical placebo on efficacy variables after 12 weeks. It includes mean baseline scores for different efficacy variables and the comparison between diclofenac sodium, topical placebo, and topical vehicle. The study assessed WOMAC pain score, WOMAC physical function, and POMA. The placebo formulation included 23% DMSO, and the vehicle formulation included 45.5% DMSO.*
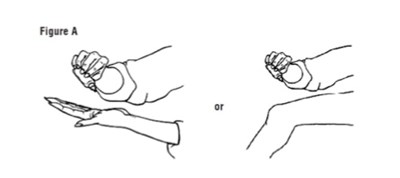
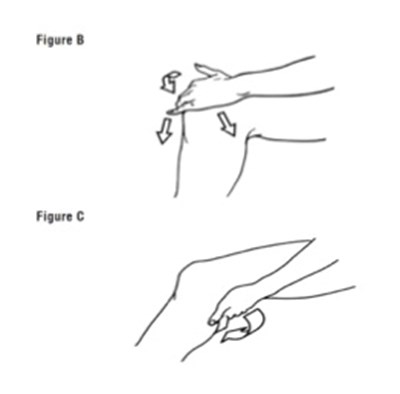
TREATMENT - Treatment Group copy

This is a list of adverse reactions associated with the topical application of Diclofenac Sodium Solution 15%. These reactions include dry skin, contact dermatitis, dyspepsin, abdominal pain, flatulence, pruritus, diarrhea, nausea, pharyngitis, edema, rash, ecchymosis, paresthesia, accidental injury, and halitosis. It is important to note that these reactions can occur both with and without application of the solution. The severity of the reactions varies, with some being more common and others less common.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.