Product Images Primaxin IV
View Photos of Packaging, Labels & Appearance
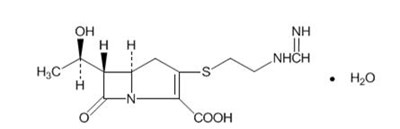
- Chemical Structure - primaxin iv 01
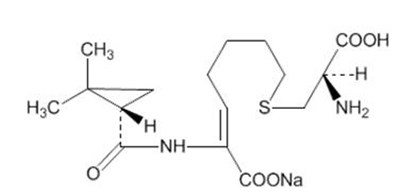
- Chemical Structure - primaxin iv 02
- PRINCIPAL DISPLAY PANEL 250 mg Bottle Label - primaxin iv 03
- PRINCIPAL DISPLAY PANEL 500 mg Bottle Label - primaxin iv 04
- Single-Dose ADD-Vantage Vial 250 mg - primaxin iv 05
- Single-Dose ADD-Vantage Vial 500 mg - primaxin iv 06
- primaxin iv 07
Product Label Images
The following 7 images provide visual information about the product associated with Primaxin IV NDC 0006-3516 by Merck Sharp & Dohme Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
PRINCIPAL DISPLAY PANEL 500 mg Bottle Label - primaxin iv 04

This is a package of Primaxin® IV, containing 500mg of imipenem, 500mg of cilastatin, and 20mg of sodium bicarbonate used as a buffer. The solution is for intravenous use only and should not be directly infused. The usual adult dosage and preparation instructions can be found in the accompanying circular. The color change of the solution from colorless to yellow does not affect potency. The package contains a single-dose vial with a serialization barcode, expiration date, and lot number. It should be stored below 25°C. This product is manufactured by Merck Sharp & Dohme LLC, based in Rahway, NJ, and is available only with a prescription.*
Single-Dose ADD-Vantage Vial 500 mg - primaxin iv 06

This is a description of a medication called Primaxinci, which includes Imipenem 500mg and Cilastatin, for injection. It comes in a single-dose vial and is cautioned not to be used for direct infusion. The trademark of Merc S1ar & Dot Gor and the registered trademark of ABBOTT LABORATORIES Inc are also included. No other information is available.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.