Product Images Caverject
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 28 images provide visual information about the product associated with Caverject NDC 0009-7686 by Pharmacia & Upjohn Company Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure A - caverject 06

The text provides instructions on the necessary supplies required to give a CAVERJECT injection. These supplies include a sterile powder vial of CAVERJECT, a diluent vial, a 3 mL syringe, a 29 or 30 gauge one-half inch needle, a 21-27 gauge sterile needle, and two alcohol swabs. It is important to note that the diluent vial and syringe are not supplied with CAVERJECT and must be obtained separately.*
Figure B - caverject 07

This text is a list of components of a syringe, including a needle cap, needle, syringe cap, tip, barrel, and plunger. The first line appears to indicate the quantity (1 syringe) and may be related to a purchase order or inventory system.*
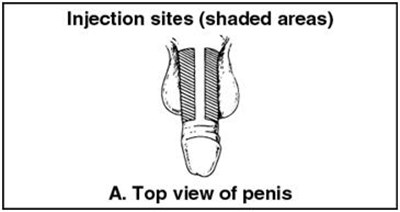
Figure P - caverject 21

This is a warning message that requests to not inject near the areas specified. The context and reasons behind the warning are not available.*
Image not available - 5 mcg - caverject 25

Caverject® alprostadil for injection is a prescription medication for intracavernosal use only. It comes in a single-dose vial with an NDC number of 0009-3701-08. The solution should be stored at a temperature between 20°C to 25°C (68°F to 77°F), and once reconstituted, it should be used within 24 hours when stored below 25°C (77°F) and not refrigerated or frozen. The dosage and use information can be found in the accompanying prescribing information. To prepare the solution, add 1 mL of diluent (bacteriostatic water for injection preserved with 0.945% w/v benzyl alcohol) and swirl until the powder dissolves. The resulting concentration is 20 meg/mL, with a total of 23.2 mcg of alprostadi and 20 mg of deliverable alprostad.*
Image not available - 10 mcg - caverject 26

This is a pharmaceutical product with the brand name Caverject IR. It contains alprostadil, which is used for intracavernosal injection. The package comes with six single-dose vials. The reconstituted solution should be used within 24 hours and stored at room temperature. The specific dosage and usage instructions are provided. The product is manufactured by Phamasia & Upjohn Co. Division of PAze I, HY, Y 10017.*
PRINCIPAL DISPLAY PANEL - 20 mcg Vial Label - caverject 27

This is a description for an injectable medication called Caverject®. The medication contains alprostadil and is used for intracavernosal use only. The accompanying prescribing information must be referred to for proper dosage and use. The medication is provided in a single-dose vial and must be stored between 2°C to 8°C (36°F to 46°F) until dispensed. After dispensation, the vials can be stored at or below 25°C (77°F) for up to 3 months or until the expiration date. When reconstituted with bacteriostatic water for injection, the resulting concentration is 40 meg/mL, providing 46.4 mcg of alprostadil. It is not recommended to use a reconstituted solution once it has been stored for 24 hours, refrigerated, or frozen.*
Principal Display Panel - 20 mcg Vial Package - caverject 28

This is a pharmaceutical product information sheet for Caverject, a drug used for intracavernosal injection in the treatment of erectile dysfunction. The sheet provides information on dosage, storage, and reconstitution of the product and states that it needs to be used under medical supervision. The text also includes the manufacturer's name and location as well as a unique product identification number.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.