Product Images Qulipta
View Photos of Packaging, Labels & Appearance
- The active ingredient of TRADENAME is atogepant, a calcitonin gene-related peptide (CGRP) receptor antagonist. The chemical name of atogepant is (S)-N-((3S,5S,6R)-6-methyl-2-oxo-1-(2,2,2-trifluoroethyl)-5-(2,3,6-trifluorophenyl)piperidin-3-yl)-2'-oxo-1',2',5,7-tetrahydrospiro[cyclopenta[b]pyridine-6,3'-pyrrolo[2,3-b]pyridine]-3-carboxamide and has the following structural formula: - qulipta 01
- Figure 1 - qulipta 02
- Figure 2 - qulipta 03
- DiagramDescription automatically generated - qulipta 04
- Chart, bar chartDescription automatically generated - qulipta 05
- PRINCIPAL DISPLAY PANELNDC 0074-7095-30QULIPTA™(atogepant) tablets10 mgRx OnlyContains 30 Tablets - qulipta 06
- PRINCIPAL DISPLAY PANELNDC 0074-7096-30QULIPTA™(atogepant) tabletsRx OnlyContains 30 Tablets30 mg - qulipta 07
- PRINCIPAL DISPLAY PANELNDC 0074-7094-30QULIPTA™(atogepant) tabletsRx OnlyContains 30 Tablets60 mg - qulipta 08
- qulipta 09
- qulipta 0a
Product Label Images
The following 10 images provide visual information about the product associated with Qulipta NDC 0074-7096 by Abbvie Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
The active ingredient of TRADENAME is atogepant, a calcitonin gene-related peptide (CGRP) receptor antagonist. The chemical name of atogepant is (S)-N-((3S,5S,6R)-6-methyl-2-oxo-1-(2,2,2-trifluoroethyl)-5-(2,3,6-trifluorophenyl)piperidin-3-yl)-2'-oxo-1',2',5,7-tetrahydrospiro[cyclopenta[b]pyridine-6,3'-pyrrolo[2,3-b]pyridine]-3-carboxamide and has the following structural formula: - qulipta 01
![qulipta-01.jpg The active ingredient of TRADENAME is atogepant, a calcitonin gene-related peptide (CGRP) receptor antagonist. The chemical name of atogepant is (S)-N-((3S,5S,6R)-6-methyl-2-oxo-1-(2,2,2-trifluoroethyl)-5-(2,3,6-trifluorophenyl)piperidin-3-yl)-2'-oxo-1',2',5,7-tetrahydrospiro[cyclopenta[b]pyridine-6,3'-pyrrolo[2,3-b]pyridine]-3-carboxamide and has the following structural formula: - qulipta 01](https://ndclist.com/assets/spl/images/8c8ab8f4-32bd-497a-befa-70c8a51d8d52/400x-qulipta-01.jpg)
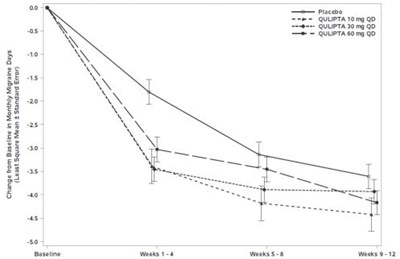
Figure 1 - qulipta 02

The text provides a table showing changes in monthly migraine days over a period of 312 weeks, including baseline and weeks 1-4, 8, and 12. It also includes the least square mean and standard error. Therefore, it can be useful for tracking the frequency of migraine headaches and potential treatment efficacy.*
Figure 2 - qulipta 03
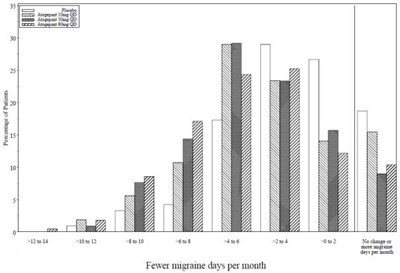
This text suggests that there is a decrease in the number of days per month that an individual experiences migraines.*
PRINCIPAL DISPLAY PANELNDC 0074-7095-30QULIPTA™(atogepant) tablets10 mgRx OnlyContains 30 Tablets - qulipta 06

QULIPTA (atogepant) tablets is a prescription drug to be used under medical supervision only. The package contains 30 tablets, and its recommended dosage can be found in the prescribing information. The drug should be stored at a temperature between 20°C and 25°C (68°F and 77°F), and it is a trademark of Allergan Pharmaceuticals International Limited. This drug is for adult use only, and it should be kept out of the reach of children.*
PRINCIPAL DISPLAY PANELNDC 0074-7096-30QULIPTA™(atogepant) tabletsRx OnlyContains 30 Tablets30 mg - qulipta 07
This is a medication package of QUUIPTA, containing 30 tablets of alagepant with 30mg strength. It is manufactured in China and distributed by the Forest Laboratories Ireland Ltd. The GTIN, lot number, expiration date, and serial number are provided. It is to be stored between 20°C and 25°C, with excursions permitted between 15°C and 30°C. The product is only available through a prescription. The package also displays the trademark of Allergan Pharmaceuticals International L, an AbbVie company.*
PRINCIPAL DISPLAY PANELNDC 0074-7094-30QULIPTA™(atogepant) tabletsRx OnlyContains 30 Tablets60 mg - qulipta 08

qulipta 09

This is a product description for QULIPTA tablets, which are intended for use as medication. The tablets are produced in China and distributed by Forest Laboratories Ireland Ltd. The packaging contains 30 tablets, with dosage information and recommended storage temperature provided. The drug is only available with a prescription (Rx Only) and contains atogepant as an active ingredient. There is also information about the manufacturer and legal trademark ownership.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.