Product Images Torpenz
View Photos of Packaging, Labels & Appearance
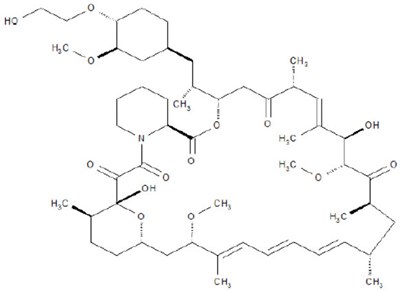
- Chemical Structure - torpenz 01
- Figure 1 - torpenz 02
- Principal Display Panel - 2.5 mg Tablet Bottle Label - torpenz 03
- Principal Display Panel - 5 mg Tablet Bottle Label - torpenz 04
- Principal Display Panel - 7.5 mg Tablet Bottle Label - torpenz 05
- Principal Display Panel - 10 mg Tablet Bottle Label - torpenz 06
Product Label Images
The following 6 images provide visual information about the product associated with Torpenz NDC 0245-0823 by Upsher-smith Laboratories, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1 - torpenz 02

This text provides information on a study comparing Everolimus 10mg with exemestane to Placebo with exemestane in terms of hazard ratio, Kaplan-Meier medians, Logrank p-value, and censoring times. The hazard ratio is 0.45 with a 95% confidence interval. Kaplan-Meier medians show 7.8 months for Everolimus 10mg with exemestane and 32 months for Placebo with exemestane. The Logrank p-value is less than 0.0001. Censoring times and sample sizes for each group are also mentioned.*
Principal Display Panel - 2.5 mg Tablet Bottle Label - torpenz 03

This text provides information on a medication named Torpenz containing Everolimus 2.5 mg tablets manufactured by Upsher-Smith Laboratories. The tablets are made in India and should be stored at 20° to 25°C to protect them from light and moisture. The package is child-resistant, and it is recommended to keep the tablets in the original container out of reach of children. The usual dosage instructions are provided in the package insert. The text also includes batch serialization coding details and expiration date information.*
Principal Display Panel - 5 mg Tablet Bottle Label - torpenz 04
This text provides information about a medication called Everolimus, USP 5 mg, manufactured by Upsher-Smith Laboratories, LLC in India. The packaging includes instructions for oral use, usual dosage guidance, storage conditions (20° to 25°C), and precautions to protect the tablets from light and moisture. The package is child-resistant and contains 30 tablets. Additionally, it includes batch serialization coding details like GTIN, SN, EXP, and LOT numbers.*
Principal Display Panel - 7.5 mg Tablet Bottle Label - torpenz 05

This text is a label for a medication manufactured for Torpenz containing Everolimus 7.5 mg in each tablet. The medication is for oral use, and the usual dosage instructions can be found in the package insert. The tablets come in a package of 30 and are prescription-only. It is advised to store the medication in the original container at 20°C to 25°C, with excursions permitted to 15°C to 30°C and to protect it from light and moisture. The package is child-resistant, and it should be kept out of reach of children. The label also includes information on batch serialization coding and expiration date.*
Principal Display Panel - 10 mg Tablet Bottle Label - torpenz 06

This text provides information about a medication called Everolimus, with a dosage of 10 mg per tablet. It includes details on how to use the medication orally, storage instructions, and precautions such as keeping it in the original container, protecting it from light and moisture, and keeping it out of reach of children. The tablets are packaged in a child-resistant package and manufactured for Torpenz by Upsher-Smith Laboratories in India. The package also includes details on the product's trademark, manufacturing details, and expiration date.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.