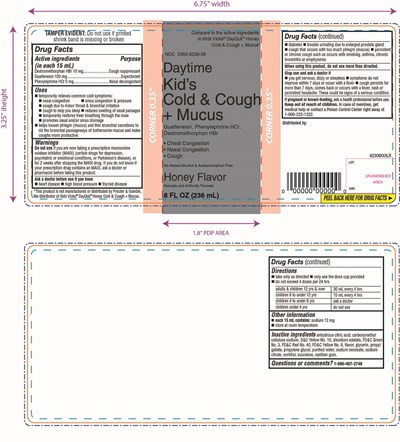
FDA Label for Walgreen Daytimel Kids Cold Cough Plus Mucus
View Indications, Usage & Precautions
- OTHER
- ACTIVE INGREDIENTS (IN EACH 15 ML)
- PURPOSE
- USES
- DO NOT USE
- ASK A DOCTOR BEFORE USE IF YOU HAVE
- OTC - WHEN USING
- STOP USE AND ASK A DOCTOR IF
- OTC - PREGNANCY OR BREAST FEEDING
- OTC - KEEP OUT OF REACH OF CHILDREN
- OVERDOSE WARNING
- DIRECTIONS
- OTHER INFORMATION
- INACTIVE INGREDIENTS
- QUESTIONS?
- PRINCIPAL DISPLAY PANEL - 236 ML BOTTLE LABEL
Walgreen Daytimel Kids Cold Cough Plus Mucus Product Label
The following document was submitted to the FDA by the labeler of this product Walgreens Co.. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Other
Drug Facts
Distributed by:
Active Ingredients (In Each 15 Ml)
Dextromethorphan HBr 10 mg
Guaifenesin 100 mg
Phenylephrine HCl 5 mg
Purpose
Cough suppressant
Expectorant
Nasal decongestant
Uses
▪ temporarily relieves common cold symptoms:▪ nasal congestion▪ sinus congestion & pressure▪ cough due to minor throat & bronchial irritation▪ cough to help you sleep▪ reduces swelling of nasal passages▪ temporarily restores freer breathing through the nose▪ promotes nasal and/or sinus drainage▪ helps loosen phlegm (mucus) and thin bronchial secretions to rid the bronchial passageways of bothersome mucus and make coughs more productive..
Do Not Use
if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Ask A Doctor Before Use If You Have
▪ heart disease▪ high blood pressure▪ thyroid disease▪ diabetes▪ trouble urinating due to enlarged prostate gland▪ cough that occurs with too much phlegm (mucus)▪ persistent or chronic cough such as occurs with smoking, asthma, chronic bronchitis or emphysema
Otc - When Using
When using this product, do not use more than directed.
Stop Use And Ask A Doctor If
▪ you get nervous, dizzy or sleepless▪ symptoms do not improve within 7 days or occur with a fever▪ cough persists for more than 7 days, comes back or occurs with a fever, rash or persistent headache
These could be signs of a serious condition.
Otc - Pregnancy Or Breast Feeding
If pregnant or breast-feeding, ask a health professional before use.
Otc - Keep Out Of Reach Of Children
Keep out of reach of children.
Overdose Warning
In case of overdose, get medical help or contact a Poison Control Center right away at 1-800-222-1222.
Directions
▪ take only as directed▪ use dose cup provided▪ do not exceed 4 doses per 24 hrs
adults & children 12 yrs & over 30 mL every 4 hrs
children 6 to under 12 yrs 15 mL every 4 hrs
children 4 to under 6 yrs ask a doctor
children under 4 yrs do not use
Other Information
▪ each 15 mL contains: sodium 12 mg▪ store at room temperature
Inactive Ingredients
citric acid, carboxymethyl cellulose sodium, D&C Yellow No. 10, disodium edetate, FD&C Green No. 3, FD&C Red No. 40, FD&C Yellow No. 6, flavor, glycerin, propyl gallate, propylene glycol, purified water, sodium benzoate, sodium citrate, sorbitol, sucralose, xanthan gum
Questions?
1-866-467-2748
Principal Display Panel - 236 Ml Bottle Label
Compare to the active ingredients in Kids Vicks ® DayQuil™ Honey Cold & Cough + Mucus
NDC 0363-6238-08
Daytime
Kid’s
Cold & Cough + Mucus
Guaifenesin, Phenylephrine HCl, Dextromethorphan HBr
• Chest Congestion• Nasal Congestion• Cough
No Added Alcohol & Acetaminophen free
Honey Flavor
Naturally and Artificially Flavored
8 FL OZ (236 mL)
*This product is not manufactured or distributed by Procter & Gamble, the distributor of Kids Vicks ® DayQuil™ Honey Cold & Cough + Mucus.
* Please review the disclaimer below.