Product Images Lamotrigine
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 10 images provide visual information about the product associated with Lamotrigine NDC 16714-893 by Northstar Rx Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
container1 - container1

This is a drug information label that contains details about Lamotrigine tablets. The tablets are in 25mg dosage, and it is recommended to take one tablet once daily. The tablet is extended-release, and each contains Lamotrigine USP 25 mg. The label also provides prescribing and dosing information, medication guides availability, and manufacturer details.*
container2 - container2

This is the packaging information for Lamotrigine Extended-Release tablets, USP, with a 50mg dosage. The prescription includes 30 tablets and has NDC number 16714-624-01. The medication comes with a medication guide, and pharmacists should dispense one to every patient. The product should be stored between 20°C and 25°C, and the manufacturer's contact information is provided.*
container3 - container3

This is a description of a prescription medication with the NDC code 16714-625-01, containing Lamotrigine, with an extended-release mechanism intended to be taken once a day. The tablets contained in the package are each 100 mg and come with a caution for careful handling. The package contains 30 tablets. There is also information regarding the manufacturer's code. The text is partly unclear due to the included output, particularly in the final line.*
container5 - container5

NDC 16714-893-01 is a medication that contains Lamotrigine in the form of extended-release tablets, USP. The medication is meant to be taken once a day and comes in a bottle containing 30 tablets. There is a caution that the product dispensed is the verified product, and the pharmacist is instructed to provide the medication guide to each patient. The rest of the text is not readable.*
container6

This is a prescription drug with NDC number 16714-627-01. It is an extended-release tablet containing 300mg of Lamotrigine, a medication commonly used to treat seizures and bipolar disorder. It is recommended that the pharmacist dispense a medication guide accompanying each prescription. There is an indication that the drug might be used for the treatment of Sxeiomn, although this is not clear. The rest of the text is not readable.*
figure1 - figure1
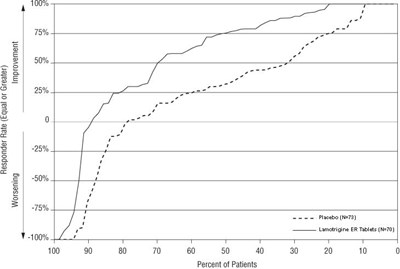
The text shows a graph presenting responder rates of patients taking Lamotrigine ER Tablets and Placebo. The horizontal axis shows the percentage of patients, and the vertical axis shows the improvement or worsening percentages in patients. The table presents percentages of patients based on the responder rate, improvement or worsening, with no available additional information on the context.*
figure2 - figure2
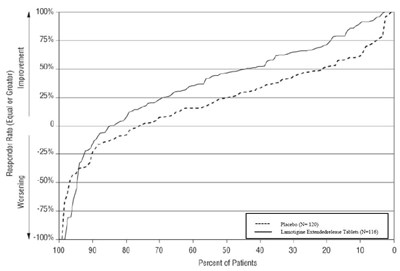
This appears to be a table showing the response rates and improvement or worsening of patients taking Lamotigine Extended-release tablets versus a placebo. The data is presented as percentages and includes different response rates (-100% to 100%). However, without more context or information about the study, the specific meaning of these percentages and response rates is not clear.*
pictures - pictures
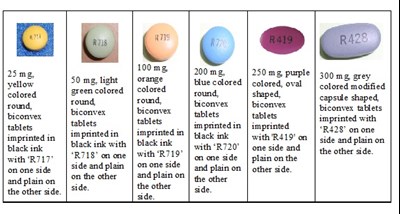
The text describes different tablets with different colors, shapes and imprints. The tablets come in 50mg, 100mg, 200mg and 250mg strengths. There is also a capsule shape tablet available in 300mg strength. The tablets are imprinted with black ink and are plain on one side.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.

