Product Images Rebif
View Photos of Packaging, Labels & Appearance
- Figure 1 - rebif 01
- Firgure A - rebif 02
- Firgure B - rebif 03
- Firgure C - rebif 04
- Firgure D - rebif 05
- Firgure E - rebif 06
- Firgure F - rebif 07
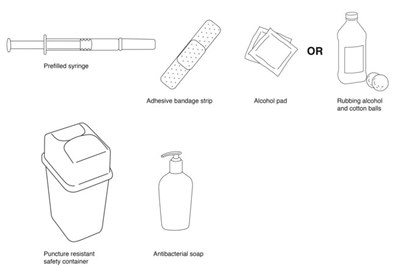
- image - rebif 07a
- Warnings - rebif 07b
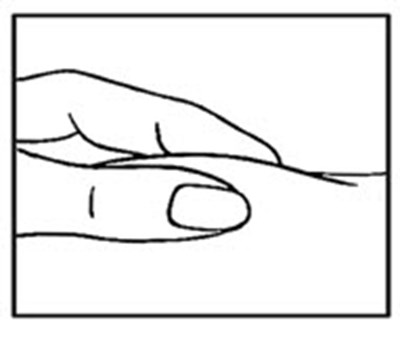
- image - rebif 07c
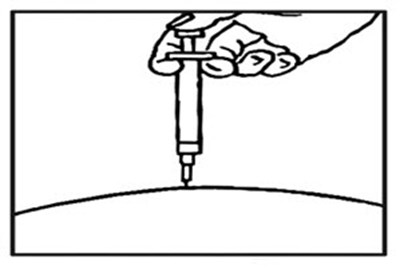
- image - rebif 07d
- image - rebif 07e
- image - rebif 07f
- Caution - rebif 07g
- image - rebif 07h
- image - rebif 07i
- image - rebif 07j
- image - rebif 07k
- PRINCIPAL DISPLAY PANEL - Kit Carton-NDC 44087-8822-1 - rebif 08
- PRINCIPAL DISPLAY PANEL - Kit Carton-NDC 44087-0188-1 - rebif 09
- PRINCIPAL DISPLAY PANEL - 12 Single Use Prefilled Syringe Carton-22 mcg/0.5 mL - rebif 10
- PRINCIPAL DISPLAY PANEL - 12 Single Use Prefilled Syringe Carton-44 mcg/0.5 mL - rebif 11
- PRINCIPAL DISPLAY PANEL - 12 Single Use Autoinjector Carton-22 mcg / 0.5 mL - rebif 12
- PRINCIPAL DISPLAY PANEL - 12 Single Use Autoinjector Carton-44 mcg / 0.5 mL - rebif 13
Product Label Images
The following 24 images provide visual information about the product associated with Rebif NDC 44087-0022 by Emd Serono, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1 - rebif 01
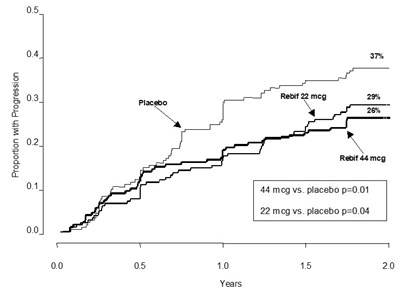
The text appears to be a graph that shows the proportion of patients who had progression over a certain number of years for a placebo group and two treatment groups (Rebif 44). The graph shows that both Rebif 44 treatment groups had a statistically significant lower proportion of patients with progression compared to the placebo group. The lower dose of Rebif 44 had a higher p-value (0.04) compared to the higher dose (0.01).*
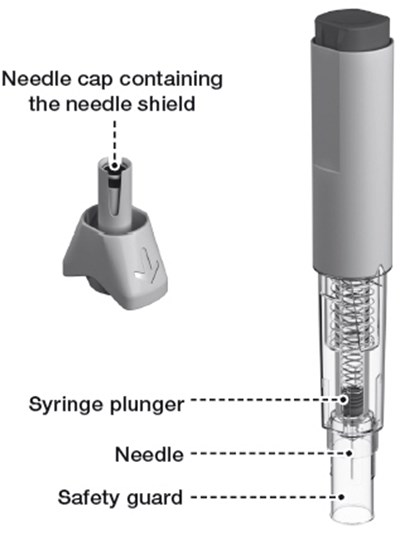
image - rebif 07c

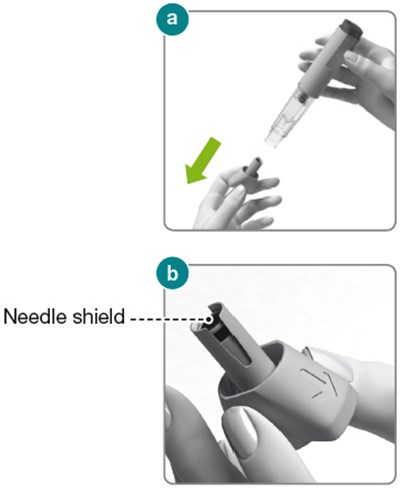
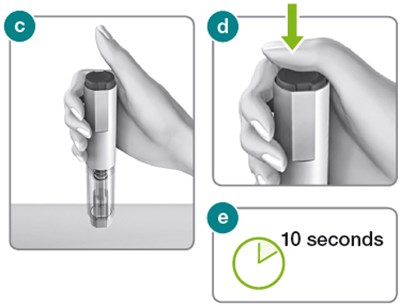
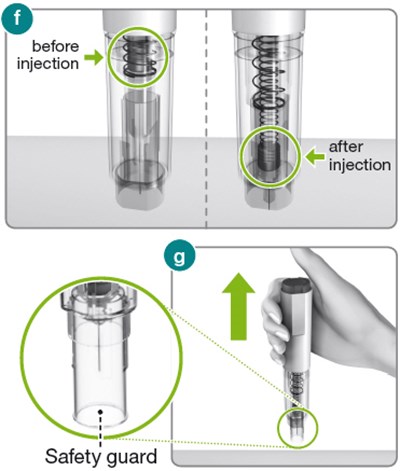
The text describes the components of an injector, including the injector button and body, the dose label area, the syringe plunger, a transparent syringe housing, and a needle cap that contains a needle shield. This information may be helpful for understanding or troubleshooting issues with the injector.*
PRINCIPAL DISPLAY PANEL - Kit Carton-NDC 44087-8822-1 - rebif 08
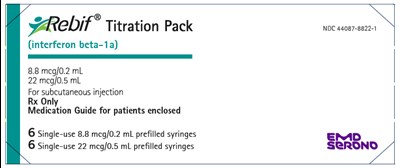
The text describes a Titration Pack for the medication %?ebif‘E (interferon beta-1a) for subcutaneous injection. The pack includes six single-use prefilled syringes in two different strengths: 8.8 mcg/0.2 mL and 22 mcg/0.5 mL. A medication guide for patients is included. The National Drug Code (NDC) is 44087-8822-1. The last line appears to be non-English characters and cannot be evaluated.*
PRINCIPAL DISPLAY PANEL - Kit Carton-NDC 44087-0188-1 - rebif 09
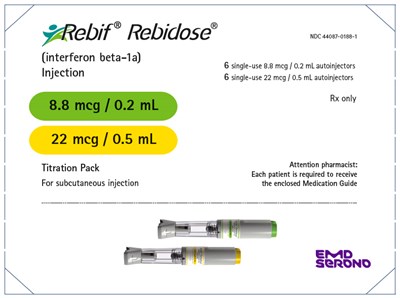
This is a medication called "yArebif Rebidose" which is an injection containing interferon beta-1a. The Titration Pack comes in two dosages, 22mcg and 0.5mL. The medication is given through subcutaneous injection and comes with a Medication Guide that must be given to each patient. The package contains 6 single-use autoinjectors of 8.8 mcg / 0.2 mL and 6 single-use autoinjectors of 22 mcg / 0.5 mL. NDC code for this is 44087-0188-1. This medication requires a prescription from a healthcare professional.*
PRINCIPAL DISPLAY PANEL - 12 Single Use Prefilled Syringe Carton-22 mcg/0.5 mL - rebif 10

AReDif is a medication that contains interferon beta-1a and is administered via subcutaneous injection. The box contains 12 prefilled syringes, each with a capacity of 0.5 mL and a concentration of 22 meg. The medication is only available with a prescription and comes with a guide for patients. The NDC code for the product is 44087-0022-3.*
PRINCIPAL DISPLAY PANEL - 12 Single Use Prefilled Syringe Carton-44 mcg/0.5 mL - rebif 11

This is a medication containing interferon beta-1a, which is intended for subcutaneous injection. It comes in a package containing 12 single-use prefilled syringes and is available only by prescription. The enclosed medication guide is provided for patient information. The NDC number is 44087-0044-3.*
PRINCIPAL DISPLAY PANEL - 12 Single Use Autoinjector Carton-22 mcg / 0.5 mL - rebif 12

Arebif® Rebidose® is an injection of interferon beta-1a provided in 12 single-use autoinjectors with a volume of 0.5 mL and a concentration of 22 μg/0.5 mL. It is intended for subcutaneous injection. A medication guide is enclosed and must be given to each patient by the pharmacist.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.