Product Images Tramadol Hydrochloride
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 7 images provide visual information about the product associated with Tramadol Hydrochloride NDC 47335-537 by Sun Pharmaceutical Industries, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
spl-tramadol-100mg - spl tramadol 100mg

This is a description for a medication with the NDC code 47335-531-83. It is an extended-release tramadol hydrochloride tablet that should be swallowed whole, not crushed or chewed. The medication guide should be dispensed with each bottle of 30 tablets. The date on the label is December 2016.*
spl-tramadol-200mg - spl tramadol 200mg

Each tablet of tramadol hydrochloride contains 200mg of the drug in an extended-release coated form. It is prescribed according to the accompanying literature and must be dispensed in a tightly sealed and light-resistant expiry-proof container. This medication should be kept out of reach of children and should be stored at a 20°C to 25°C temperature range. These tablets are manufactured by Sun Pharmaceutical Industries, Inc. and should not be split, crushed, or chewed. The medication guide provided separately to each patient should be handed over. No additional information is available.*
spl-tramadol-300mg - spl tramadol 300mg

This is a medication for pain relief called Tramadol Hydrochloride. It comes in the form of extended-release coated tablets, where each tablet has 300 mg of tramadol hydrochloride, USP. It is essential to read the prescribing literature for the usual dosage. The tablets must be swallowed whole with liquid and not cut, crushed, or chewed. The pharmacist should dispense the medication guide separately to each patient. It must be stored in a tight, light-resistant container, away from children, between 20° to 25°C (68° 0 77°F), with excursions between 15° and 30°C (59° and 86°F). It is distributed by Sun Pharmaceutical Industries, Inc., and manufactured by Sun Pharmaceutical Industries Ltd. located in India.*
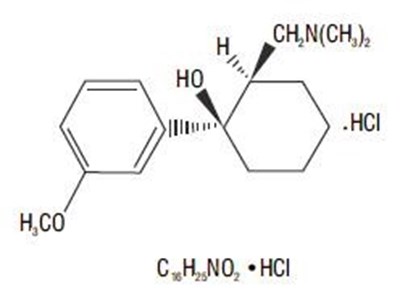
spl-tramadol-figure2 - spl tramadol figure2
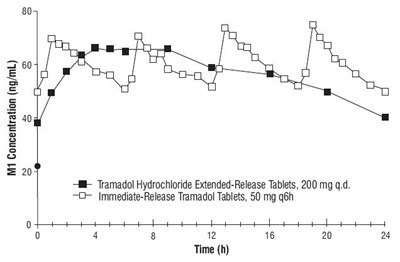
This appears to be a graph showing the concentration of M1 in ng/mL over time (in hours) for a patient taking Tramadol Hydrochloride Extended-Release Tablets (at a dose of 200mg per day) and Immediate-Release Tramadol Tablets (at a dose of 50mg every 6 hours). Beyond that, no further context is available.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.