Product Images Dengvaxia
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 7 images provide visual information about the product associated with Dengvaxia NDC 49281-605 by Sanofi Pasteur Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
PRINCIPAL DISPLAY PANEL - Kit Carton - dengvaxia 05
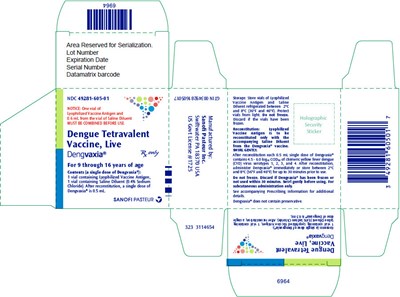
This appears to be a label for a vaccine vial, containing information about the lot number, expiration date, serial number, and a datamatrix barcode. The label also includes instructions for use, with warnings about not using the vaccine if it has been frozen or left out for more than 30 minutes, and details about the contents of the vial. The vaccine is identified as "Dengvaxia®", which appears to be a tetravalent dengue vaccine produced by Sanofi Pasteur.*
PRINCIPAL DISPLAY PANEL - 0.5 mL Vial Label - dengvaxia 06

This is a product description for Dengvaxia, a vaccine manufactured by Sanofi Pasteur Inc. The package contains a single dose of 0.5 mL, intended for subcutaneous use after reconstitution. The product is identified by its NDC code, 49281-606-58, and the text indicates that it is for sale to authorized personnel only.*
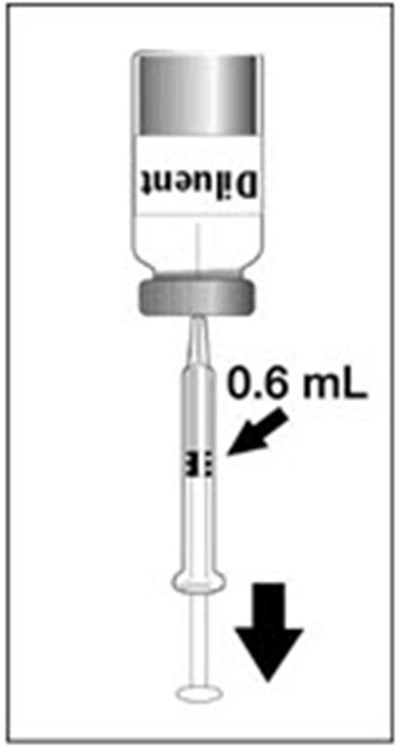
PRINCIPAL DISPLAY PANEL - 0.6 mL Vial Label - dengvaxia 07

This is a product label for a vaccine diluent called Saline Diluent, produced by Sanofi Pasteur Inc. The diluent is meant to be added to a vial of a lyophilized vaccine antigen called B2ty to create the vaccine Dengvaxia. The label contains the vaccine's National Drug Code (NDC) and a warning that the diluent should not be used alone. There is also a manufacturing code and location provided.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.