Product Images Tecentriq Hybreza
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 9 images provide visual information about the product associated with Tecentriq Hybreza NDC 50242-933 by Genentech, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 2 - tecentriq 02
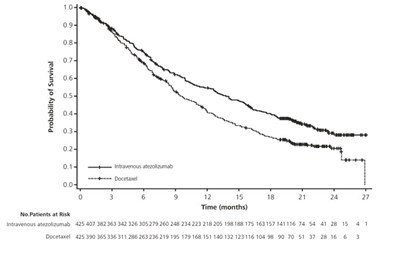
This data appears to be related to a survival analysis study where the number of individuals at risk over time is recorded. The table shows the number of individuals at risk for each time point in months, along with a group that received chemotherapy. This information can be used to analyze the survival rates and outcomes of individuals undergoing chemotherapy compared to those who did not.*
Figure 4 - tecentriq 04

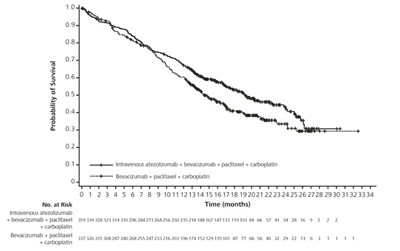
This is a data table showing the probability of survival for different treatment options over a period of months. The data includes the number of participants at risk and the time in months, with differences in survival rates observed between treatments such as intravenous atezolizumab with pociase protein-bound and carboplatin, as well as pacitaxel protein-bound with carboplatin. The information suggests a comparison of survival outcomes based on the treatments administered.*
Figure 7 - tecentriq 07
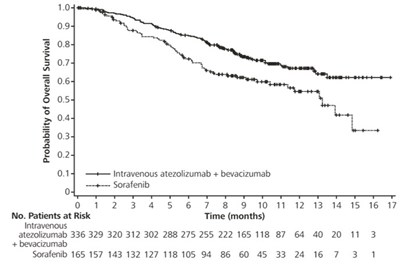
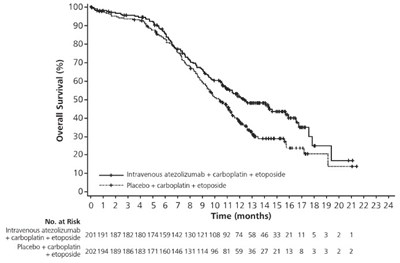
This is a table showing the probability of overall survival for patients receiving the combination treatment of intravenous atezolizumab + bevacizumab with Sorafenib over a period of 17 months. The data includes percentages of survival at different time points, ranging from 1 to 17 months. This information can be helpful for evaluating the efficacy and potential outcomes of these treatment options for patients.*
Figure 8 - tecentriq 08

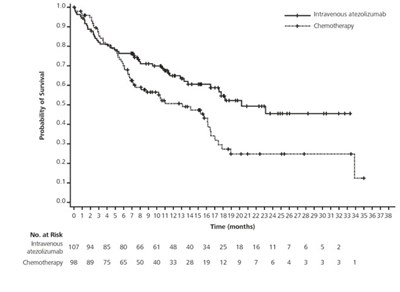
This is a table displaying the number of patients remaining at risk over time (in months) for two different treatments: Intravenous atezolizumab and cobimetinib + vemurafenib. The data also includes the percentage of patients at each timepoint.*
PRINCIPAL DISPLAY PANEL - 15 mL Vial Box - tecentriq 09

This text provides information about a medication called Tecentriq Hybreza which is an injection with the active ingredients atezolizumab and hyaluronidase-tgjs, intended for subcutaneous use. The NDC code is 50242-933-01, and it is in a single-dose vial format. It emphasizes discarding any unused portion and advises pharmacists to give the accompanying medication guide to each patient.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.