Product Images Vitrakvi
View Photos of Packaging, Labels & Appearance
- image of 100 mg carton principal panel - 60 capsules - carton caps 100
- image of 25 mg carton principal panel - 60 capsules - carton caps 25
- image of 20 mg/mL carton principal panel - 100 mL oral solution - carton sol
- image of the chemical structure of VITRAKVI - chem struct
- image of supplies needed to take or give a dose of VITRAKVI oral solution - fig a
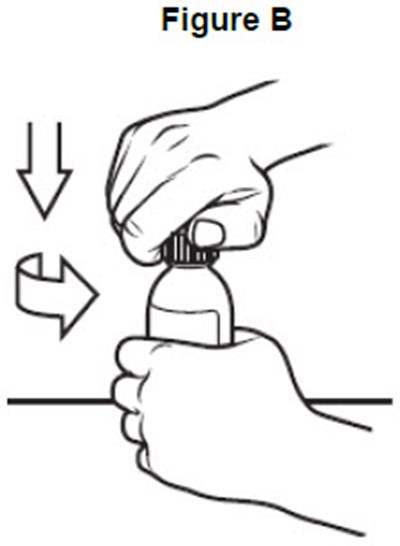
- image of how to open the bottle - fig b
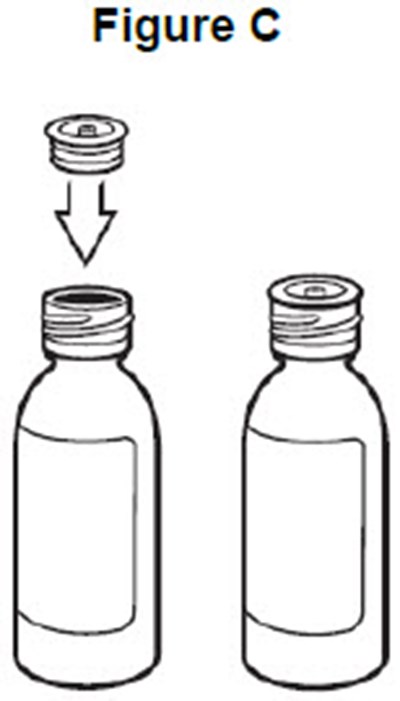
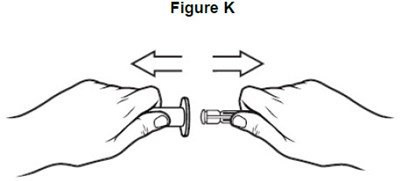
- image of how to insert the adaptor into the bottle - fig c
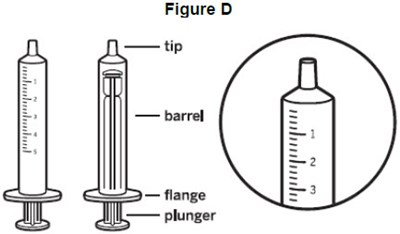
- image of the oral syringe and a close up of the markings in mL - fig d
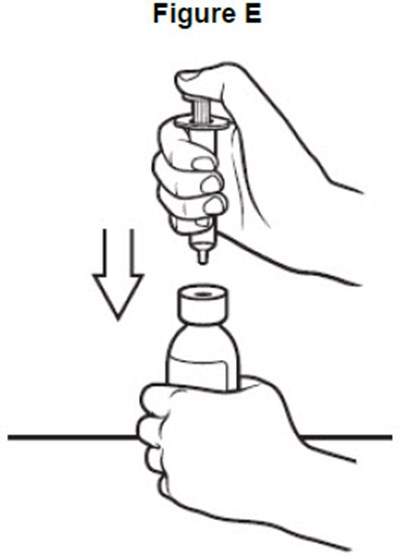
- image of of how to insert the oral syringe into the bottle - fig e
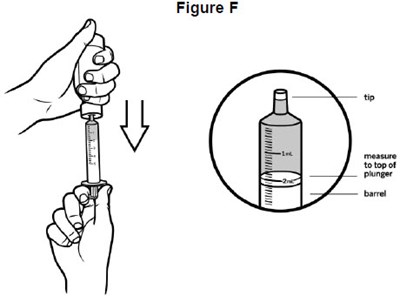
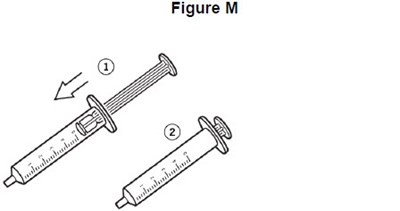
- image of how to draw VITRAKVI into the oral syringe for proper dose - fig f
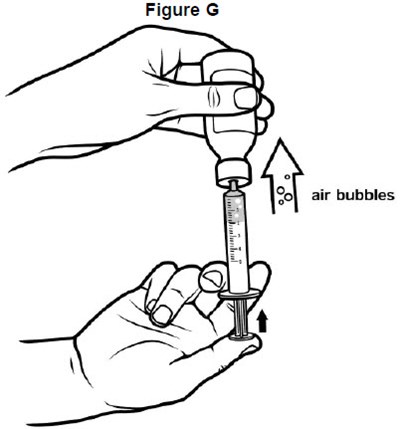
- image of how to expell air bubbles in the oral syringe - fig g
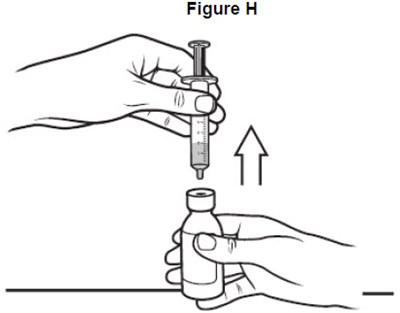
- image of how to remove the oral syringe from the bottle - fig h
- image of how to properly press the plunger to deliver the dose - fig i
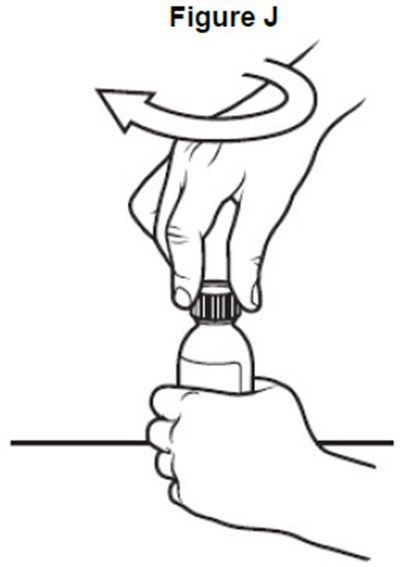
- image of recapping the bottle - fig j
- image of removing the plunger from the barrel of the oral syringe - fig k
- image of rinsing the oral syringe (plunger and barrel) - fig l
- image of re-insertion of plunger into oral syringe barrel - fig m
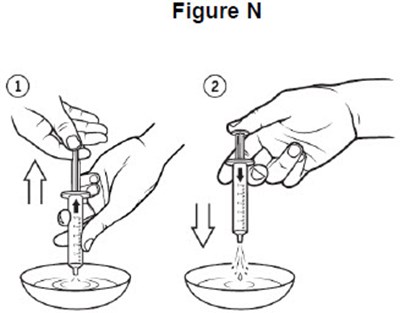
- image of cleaning oral syringe using warm water - fig n
- image of rinsing the plunger and barrel of the oral syringe a 2nd time - fig o
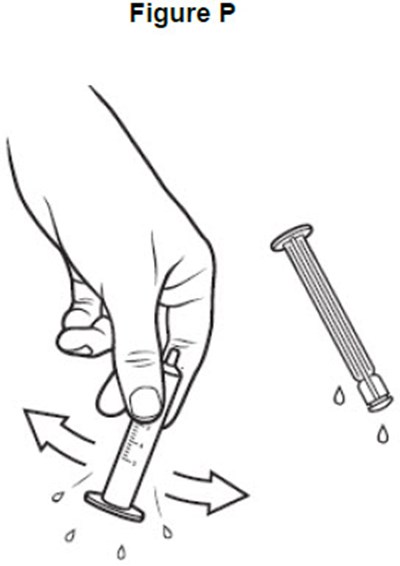
- image of shaking excess water from the barrel and plunger - fig p
- image of where to write the date of when the bottle was opened - fig q
- image of 100 mg label principal panel - 60 capsules - label caps 100
- image of 25 mg label principal panel - 60 capsules - label caps 25
- image of 20 mg/mL label principal panel - 100 mL oral solution - label sol
Product Label Images
The following 24 images provide visual information about the product associated with Vitrakvi NDC 50419-390 by Bayer Healthcare Pharmaceuticals Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
image of 100 mg carton principal panel - 60 capsules - carton caps 100

VITRAKVI® capsules contain 100mg of larotrectinib sulfate equivalent to 123mg of larotrectinib. These capsules are intended for oral use and are available in a package of 60 capsules.*
image of 25 mg carton principal panel - 60 capsules - carton caps 25

VITRAKVI is an oral medication that comes in the form of capsules containing 25 mg of larotrectinib, which is equivalent to 30.7 mg of larotrectinib sulfate. This medication is intended for oral use and is available in a package of 60 capsules.*
image of 20 mg/mL carton principal panel - 100 mL oral solution - carton sol

VITRAKVI® is an oral solution medication containing 20mg/mL of larotrectinib. Each bottle has a total of 100mL of the solution, equivalent to 24.6mg/mL of larotrectini sulfate. This medication is intended for oral use.*
image of supplies needed to take or give a dose of VITRAKVI oral solution - fig a

The text contains a description of medical equipment including an oral syringe with a capacity of 5mL, a bottle and a bottle adaptor. A figure labeled as A is present.*
image of 100 mg label principal panel - 60 capsules - label caps 100

This is a prescription medication with the brand name VITRAKVI®. The drug comes in capsule form and each capsule contains 100mg of the active ingredient called Larotrectinib. The recommended dosage is provided within the prescribing information. The medication is packaged in a bottle containing 60 capsules and should be kept away from children. It is to be stored at a temperature range of 20°C to 25°C (68°F to 77°F), with permitted excursions ranging from 15°C to 30°C (59°F to 86°F). The manufacturer of this medication is Bayer HealthCare Pharmaceuticals Inc.*
image of 25 mg label principal panel - 60 capsules - label caps 25

This text provides information about VITRAKVI®, which is a capsule medication with the active ingredient Larotrectinib. It also mentions the NDC code (50419-390-01) and provides some precautions for use. There is a statement about keeping it out of reach of children and the recommended storage temperature. The text indicates that prescription information should be consulted for further details. The manufacturer is identified as BayrHeathCoePamacuticss, located at 7381 sasesers.*
image of 20 mg/mL label principal panel - 100 mL oral solution - label sol

NDC 50419-392-01 is a medication called VITRAKVI® in the form of an oral solution with a concentration of 20 mg/mL. However, no further information is available in the text provided.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.