FDA Label for Calm Eczema Therapy Wash
View Indications, Usage & Precautions
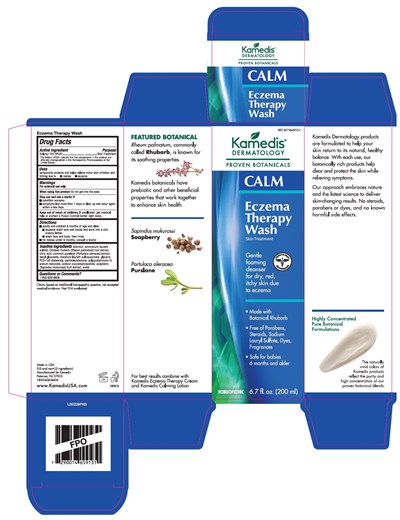
Calm Eczema Therapy Wash Product Label
The following document was submitted to the FDA by the labeler of this product Kamedis. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Drug Facts
Active Ingredient
Sulphur 12X HPUS*
* The letters ‘HPUS’ indicate that the components in this product are officially monographed in the Homeopathic Pharmacopoeia of the United States
Purpose
Skin Treatment
Use
- Temporarily protects and helps relieve minor skin irritation and itching due to:
- rashes
- eczema
Warnings
For external use only
When Using This Product
- Do not get into eyes
Stop Use And Ask A Doctor If
- Condition worsens
- Symptoms last more than 7 days or clear up and occur again within a few days
Keep Out Of Reach Of Children
- If swallowed, get medical help or contact a Poison Control Center right away
Directions
- Apply as needed
Inactive Ingredients
Allantoin, Ammonium Laureth Sulfate, Chinese Rhubarb (Rheum Palmatum) Root Extract, Citric Acid, Common Purslane (Portulaca Oleracea) Extract, Decyl Glucoside, Disodium Laureth Sulfosuccinate, Glycerin, PEG-150 Distearate, Pentadecalactone, Polyquaternium-10, Sodium Benzoate, Sodium Cocoamphoacetate, Soapberry (Sapindus Mukurossi) Fruit Extract, Water.
Questions Or Comments?
1-855-626-6606
Carton Label
* Please review the disclaimer below.