Product Images Esmolol Hydrochloride
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 9 images provide visual information about the product associated with Esmolol Hydrochloride NDC 51662-1371 by Hf Acquisition Co Llc, Dba Healthfirst, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
ADVERSE - Adverse

This is a description of the frequency of different medical conditions and reactions during the use of esmolol hydrochloride infusion. The text identifies the frequency of hypotension, both asymptomatic and symptomatic, as well as infusion site reactions, nausea, dizziness, and somnolence. It also notes that hypotension resolves for most patients during the infusion or within 30 minutes after discontinuation.*
DESCRIPTION - Description

Esmolol Hydrochloride Injection is a medication that contains 10mg of Esmolol Hydrochloride per mL, along with Water for Injection, Sodium Acetate Trihydrate, Glacial Acetic Acid, Sodium Hydroxide, and Hydrochloric Acid. The medication is used to treat various medical conditions by altering the heart rate.*
DOSAGE - Dosage And Admin

This text describes different dosage options for a medication, with recommendations for optional loading doses and suggested amounts per kilogram of body weight and duration of administration. The medication name and purpose are not given.*
DOSAGE fORMS - Dosage Forms

Esmolol Hydrochloride Injection comes in a 10 mL vial containing a total dose of 100 mg. The concentration of Esmolol Hydrochloride is 10 mg/mL. This product is typically used as a short-acting beta-blocker to treat heart rhythm problems.*
HOW SUPPLIED - How Supplied

This is a product with NDC number 63323-652-10, containing 100mg per 10mL (10mg per mL) in a single dose vial of 10mL. It is available in a package of 25.*
SERIALIZED LABELING - Serialized Labeling
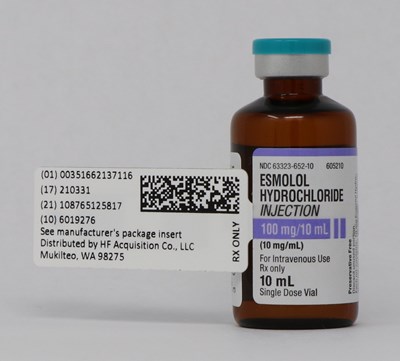
This is a description for a medicine. The name of the drug is Esmolol Hydrochloride Injection, which is only available with a prescription. The drug is distributed by HF Acquisition Co., LLC and provided in a single dose vial. The National Drug Code (NDC) is 63323-652-10 and the manufacturer's package insert should be consulted before use. The drug is intended for intravenous use and is preservative-free. The text also includes some unidentifiable characters, but this does not affect the description.*
VIAL LABEL - Vial Label

This appears to be a label for a medication called Esmolol Hydrochloride. It is a single dose vial meant for intravenous use with a concentration of 10mg/mL and a total volume of 10mL. The label also includes dosage information and instructions for use.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.

