Product Images Neupogen
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 38 images provide visual information about the product associated with Neupogen NDC 55513-209 by Amgen Inc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
neupogen 01
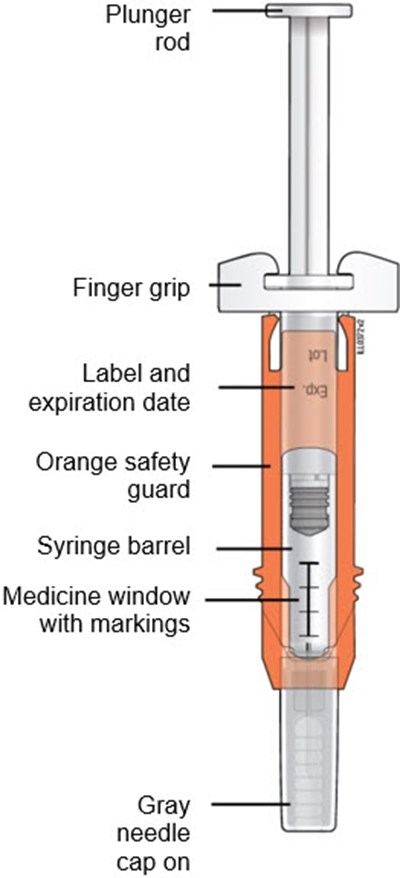
This appears to be a description of a syringe with various parts specified such as the plunger rod, finger grip, label and expiration date, orange safety guard, syringe barrel, and a medicine window with markings.*
neupogen 02
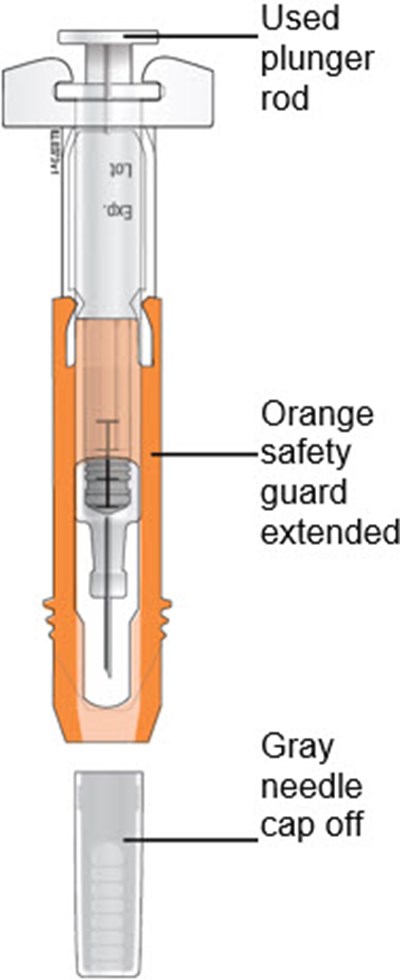
This text appears to be a list of items or components. It states "Used plunger rod," referring to a previously owned plunger rod. Without additional context, it is difficult to determine the specific application or use of this plunger rod.*
Guide to parts - After use. - neupogen 04

This appears to be a description of a medical product, likely a needle cap for a syringe. The cap is gray in color and has an expiration date window with markings to indicate when the medication inside has expired.*
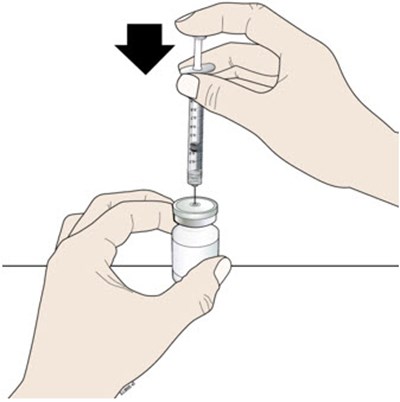
Open the tray by peeling away the cover. Grab the orange safety guard to remove the prefilled syringe from the tray. - neupogen 05

Inspect the medicine and prefilled syringe. - neupogen 06

This text provides a brief description of different body parts, including the upper abdomen and thigh.*
Alcohol wipe, Cotton ball or gauze pad, Adhesive bandage, Sharps disposal container - neupogen 08

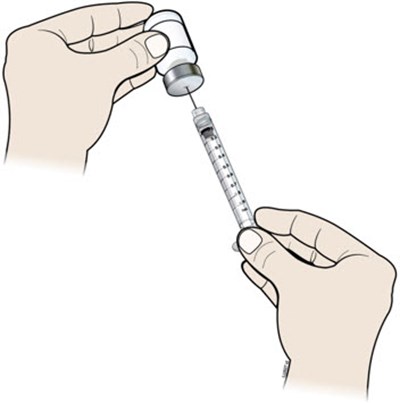
This is a description of a syringe barrel.*
Put the used prefilled syringe in a FDA-cleared sharps disposal container right away after use. Do not throw away (dispose of) the prefilled syringe in your household trash. - neupogen 14
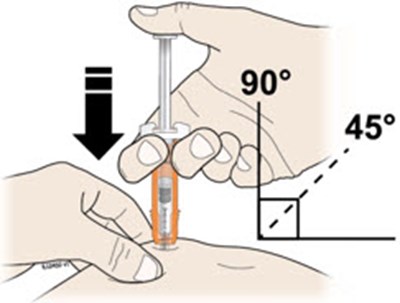
Vial Disposable syringe and needle 2 alcohol wipes Cotton ball or gauze pad Adhesive bandage Sharps disposal container - neupogen 15
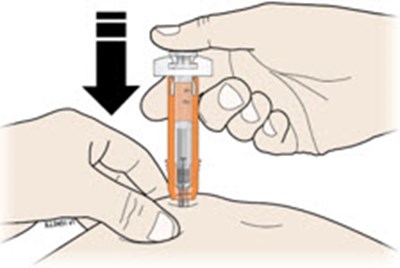
Hold the syringe by the barrel with the needle cap pointing up. Carefully pull the needle cap straight off and away from your body. - neupogen 18
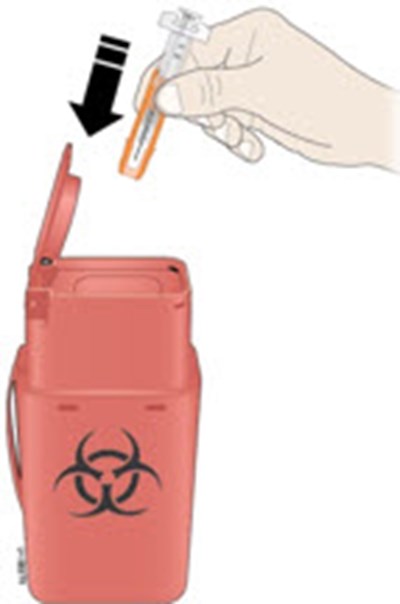
Important: Throw the needle cap into the sharps disposal container. - neupogen 19
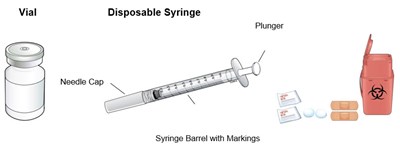
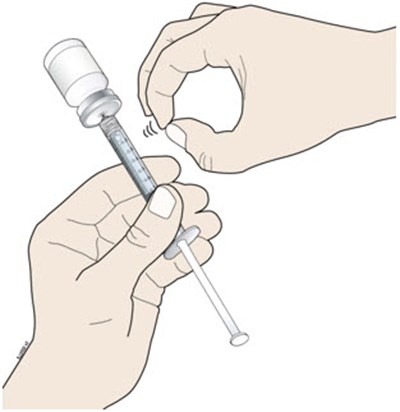
This is a description of a disposable syringe that includes a vial, plunger, and barrel with markings. The specific details of the product are not available.*
Put your used syringes, needles, and vials in a FDA-cleared sharps disposal container right away after use. Do not throw away (dispose of) needles, syringes and vials in your household trash. - neupogen 20
NDC 55513-530-10 10 – 1 mL Single Dose Vials AMGEN® NEUPOGEN® (filgrastim) for injection A Recombinant Granulocyte Colony Stimulating Factor (rG-CSF) derived from E Coli 300 mcg 300 mcg/1 mL (3 x 107 Units/1 mL) For Subcutaneous or Intravenous Use Only Sterile Solution – No Preservative Refrigerate at 2 to 8C (36 to 46F). Avoid Shaking. Amgen Inc. Thousand Oaks, CA 91320 U.S.A. U.S. License No. 1080 - neupogen 21
NDC 55513-546-10 10 – 1.6 mL Single Dose Vials AMGEN® NEUPOGEN® (filgrastim) for injection A Recombinant Granulocyte Colony Stimulating Factor (rG-CSF) derived from E Coli 480 mcg 480 mcg/1.6 mL 300 mcg/1 mL (3 x 107 Units/1 mL) For Subcutaneous or Intravenous Use Only Sterile Solution – No Preservative Refrigerate at 2 to 8C (36 to 46F). Avoid Shaking. Amgen Inc. Thousand Oaks, CA 91320 U.S.A. U.S. License No. 1080 - neupogen 22

NDC 55513-924-91 1 x 300 mcg/0.5 mL Single Dose Prefilled Syringe with 27 Gauge Needle AMGEN® NEUPOGEN® SingleJect® (filgrastim) injection A Recombinant Granulocyte Colony Stimulating Factor (rG-CSF) derived from E Coli 300 mcg Single Dose Prefilled Syringe with 27 Gauge Needle 300 mcg/0.5 mL For Subcutaneous Use Only This Product Contains Dry Natural Rubber Sterile Solution – No Preservative Refrigerate at 2 to 8C (36 to 46F). Avoid Shaking. Rx Only Manufactured by Amgen Inc. Thousand Oaks, CA 91320 U.S.A. - neupogen 23
NDC 55513-924-10 10 x 300 mcg/0.5 mL Single Dose Prefilled Syringes with 27 Gauge Needles AMGEN® NEUPOGEN® SingleJect® (filgrastim) injection A Recombinant Granulocyte Colony Stimulating Factor (rG-CSF) derived from E Coli 300 mcg Single Dose Prefilled Syringes with 27 Gauge Needles 300 mcg/0.5 mL For Subcutaneous Use Only This Product Contains Dry Natural Rubber Sterile Solution – No Preservative Refrigerate at 2 to 8C (36 to 46F). Avoid Shaking. Rx Only Manufactured by Amgen Inc. Thousand Oaks, CA 91320 U.S.A. U.S. License No. 1080 - neupogen 24
NDC 55513-209-91 1 x 480 mcg/0.8 mL Single Dose Prefilled Syringe with 27 Gauge Needle AMGEN® NEUPOGEN® SingleJect® (filgrastim) injection A Recombinant Granulocyte Colony Stimulating Factor (rG-CSF) derived from E Coli 480 mcg Single Dose Prefilled Syringe with 27 Gauge Needle 480 mcg/0.8 mL For Subcutaneous Use Only This Product Contains Dry Natural Rubber Sterile Solution – No Preservative Refrigerate at 2 to 8C (36 to 46F). Avoid Shaking. Rx Only Manufactured by Amgen Inc. Thousand Oaks, CA 91320 U.S.A. - neupogen 25
NDC 55513-209-10 10 x 480 mcg/0.8 mL Single Dose Prefilled Syringes with 27 Gauge Needles AMGEN® NEUPOGEN® SingleJect® (filgrastim) injection A Recombinant Granulocyte Colony Stimulating Factor (rG-CSF) derived from E Coli 480 mcg Single Dose Prefilled Syringes with 27 Gauge Needles 480 mcg/0.8 mL For Subcutaneous Use Only This Product Contains Dry Natural Rubber Sterile Solution – No Preservative Refrigerate at 2 to 8C (36 to 46F). Avoid Shaking. Rx Only Manufactured by Amgen Inc. Thousand Oaks, CA 91320 U.S.A. U.S. License No. 1080 - neupogen 26
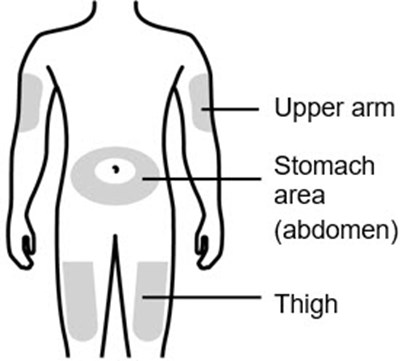
This text provides the names of three body parts, "Upper arm", "Stomach area (abdomen)", and "Thigh". It can be used as a reference for identifying specific regions of the human body.*
neupogen 27
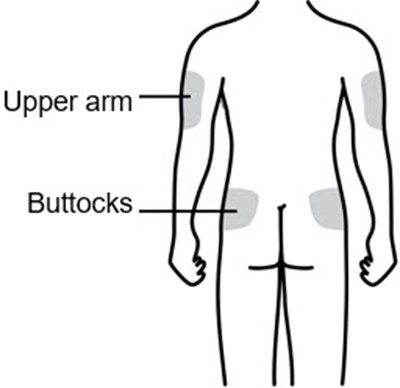
This text provides two anatomical terms, "Upper arm" and "Buttocks". It can be useful for identifying specific areas of the human body.*
neupogen 32

NEUPOGEN is a sterile solution for subcutaneous or intravenous use to stimulate granulocyte production in patients with certain types of cancer. It is a recombinant granulocyte colony-stimulating factor (G-CSF) derived from E. coli. Each vial contains 300 micrograms/mL and comes in a single-dose vial. NEUPOGEN must be stored in the refrigerator at 2°C to 8°C and should not be shaken. The manufacturer is Amgen Inc. in California, and it has been approved by the US FDA (license no. 1080).*
neupogen 33

NEUPOGEN is a sterile solution medication used for subcutaneous and intravenous use only. It is a recombinant human granulocyte colony-stimulating factor that is used to stimulate white blood cell production in the body. It is available in 10-16 mL single-dose vials, with each vial containing 480 meg/0.5 mL or 300 meg/1 mL. It is distributed by Amgen, Inc. in Thousand Oaks, California.*
neupogen 34

This is a description of a medication called Filgrastim, which is a recombinant Granulocyte Colony Stimulating Factor derived from E Coli, contained in a pre-filled syringe with a 27 gauge needle. It is intended for subcutaneous use only and contains dry natural rubber. The syringe is sterile and should be refrigerated at 2°C to 8°C, avoiding shaking. It is manufactured by Amgen Inc. located in Thousand Oaks, California, USA. The product is licensed under license number 1080.*
neupogen 35

This is a single dose pre-filled syringe with a 27-gauge needle containing 1300 mog/0.5 mL of a recombinant granulocyte colony-stimulating factor injection derived from E. coli. The syringe needs to be refrigerated between 2°C to 8°C and should not be shaken. The product is intended for subcutaneous use only and contains dry natural rubber manufactured by Amgen Inc. The solution is sterile without any preservatives present. The NDG number is 55513-924-91.*
neupogen 36

This is a description for a medication called "Nf UPOG£N" which is a single dose prefilled syringe containing a recombinant granulocyte colony stimulating factor used for subcutaneous injections. The syringes come with 27 Gauge needles, containing 480 meg/0.8 mL of solution to be refrigerated between 2°C to 8°C (36°F to 46°F). It is important to avoid shaking the syringe since it contains a sterile solution without preservatives and dry natural rubber. The medication is manufactured by Amgen Inc., with US License No. 1080.*
neupogen 37

This is a description of a medication called NEUPOGEN. The medication comes in a single-dose prefilled syringe with a 27 gauge needle. It is a recombinant granulocyte colony stimulating factor derived from E. Coli. The product should be refrigerated at 2°C to 8°C (36°F to 46°F) and should be avoided from shaking. It is for subcutaneous use only and contains dry natural rubber. It is manufactured by Amgen in Thousand Oaks, CA 91320 USA.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.













