Product Images Zomacton
View Photos of Packaging, Labels & Appearance
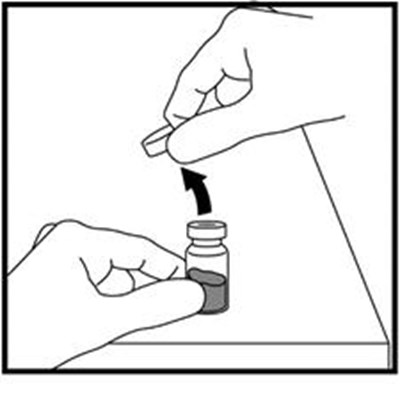
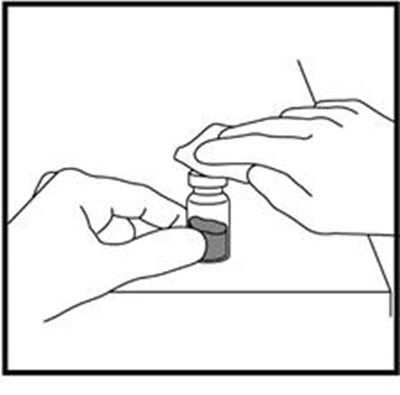
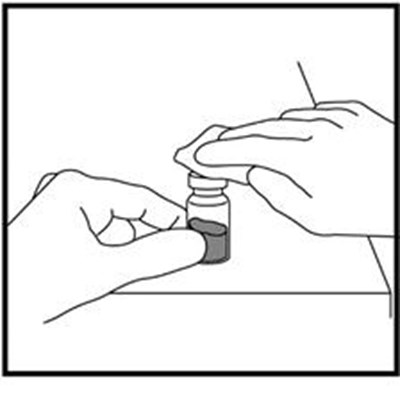
- Figure A - zomacton 01
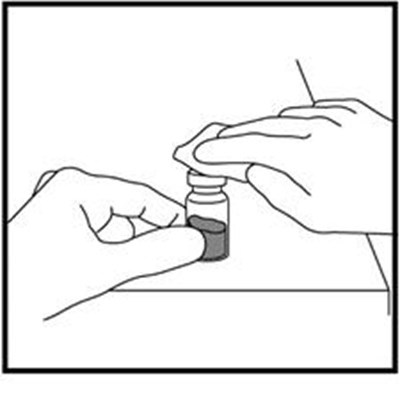
- Figure B - zomacton 02
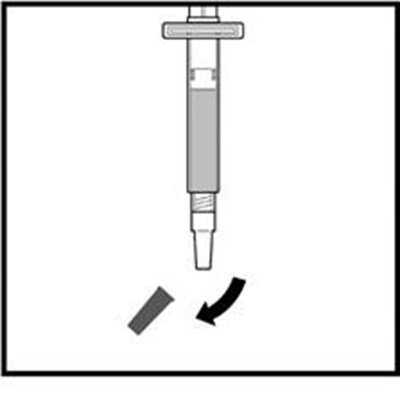
- Figure C - zomacton 03
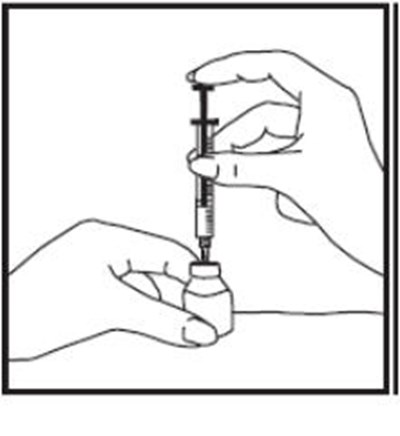
- Figure D - zomacton 04
- Figure E - zomacton 05
- Figure F - zomacton 06
- Figure G - zomacton 07
- Figure H - zomacton 08
- Figure I - zomacton 09
- Figure J - zomacton 10
- Figure K - zomacton 11
- Figure L - zomacton 12
- Figure M - zomacton 13
- Figure N - zomacton 14
- Figure O - zomacton 15
- Figure P - zomacton 16
- Figure Q - zomacton 17
- Figure R - zomacton 18
- Figure S - zomacton 19
- Figure T - zomacton 20
- Figure U - zomacton 21
- Figure V - zomacton 22
- Principal Display Panel - 5 mg Vial Label - zomacton 23
- Principal Display Panel - 5 mL Diluent Vial Label - zomacton 24
- Principal Display Panel - 5 mg Kit Carton - zomacton 25
Product Label Images
The following 25 images provide visual information about the product associated with Zomacton NDC 55566-1801 by Ferring Pharmaceuticals Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
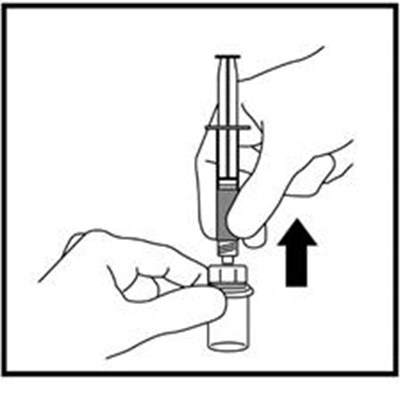
Figure B - zomacton 02

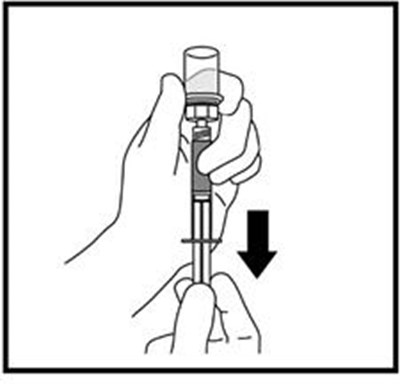
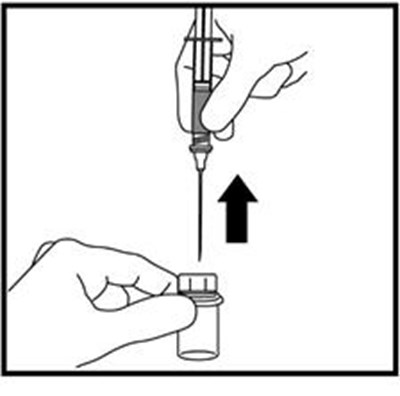
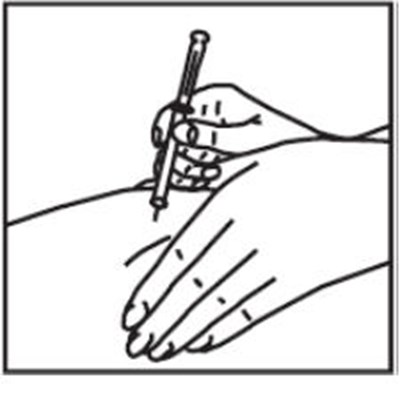
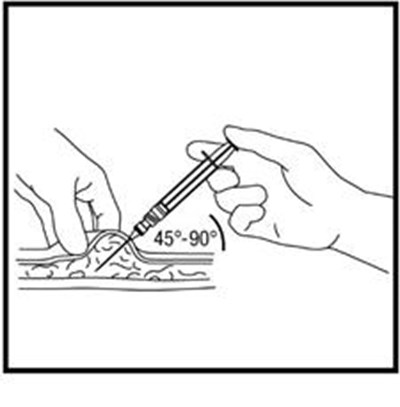
This text is providing information about a syringe with a mixing needle and a medication called ZOMACTON 10mg.*
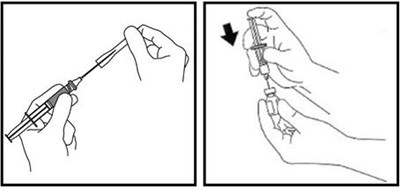
Figure C - zomacton 03

The text is not clear and contains several errors and non-English characters. Therefore, the description cannot be generated.*
Principal Display Panel - 5 mg Vial Label - zomacton 23

ZOMACTON 5mg is a medication for subcutaneous injection containing 5mg of somatropin, a growth hormone that stimulates cell regeneration and reproduction in humans, and 30mg of mannitol. Each package comes with one vial of ZOMACTON and one vial of bacteriostatic 0.9% sodium chloride injection, USP. ZOMACTON is to be reconstituted with bacteriostatic 0.9% sodium chloride injection, USP and refrigerated. The medication should not be frozen and must be used within 14 days of reconstitution. The usual dosage is contained in the package insert. The medication was made in Germany for Ferring Pharmaceuticals Inc. in the USA.*
Principal Display Panel - 5 mL Diluent Vial Label - zomacton 24

ZOMACTON 10mg (somatropin) for injection is a drug from Germany containing 10 mg of somatropin lyophilized with 10 mg of mannitol. The package includes a vial of ZOMACTON (somatropin) for Injection 10 mg, a prefilled diluent syringe, and a needle attachment. Each prefilled syringe contains 1 mL of bacteriostatic water for injection containing 0.33% m-cresol as an antimicrobial preservative. The reconstituted solution has a pH of 7.0 - 0.0. The package insert should be consulted for the usual dosage. The drug is manufactured by Ferring Pharmaceuticals, NJ, USA.*
Principal Display Panel - 5 mg Kit Carton - zomacton 25

ZOMACTON 10mg is a medication for injection containing somatropin. Each vial contains 10 mg of somatropin lyophilized with 10 mg of mannitol. The prefilled syringe contains 1 mL of Bacteriostatic water for reconstitution. The solution has a pH of 7.0 - 0.0 and contains 0.33% metacresol as an antimicrobial preservative. The usual dosage is to be determined with package insert guidelines. The medication is for subcutaneous use only and should be refrigerated at 2°C to 8°C. The reconstituted vial should be used within 28 days and kept away from light. It should only be used with the needle-free ZOMA-Jet 10. The medication is manufactured in Germany and distributed by Ferring Pharmaceuticals Inc. in Parsippany, NJ, USA.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.