FDA Label for Cetirizine Hydrochloride
View Indications, Usage & Precautions
Cetirizine Hydrochloride Product Label
The following document was submitted to the FDA by the labeler of this product Jubilant Cadista Pharmaceuticals Inc.. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Active Ingredient ( In Each Chewable Tablet )
Cetirizine Hydrochloride 5 mg
Cetirizine Hydrochloride 10 mg
Purpose
Antihistamine
Uses:
temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
runny nose
itchy, watery eyes
sneezing
itching of the nose or throat
When Using This Product:
drowsiness may occur
avoid alcoholic drinks
alcohol, sedatives and tranquilizers may increase drowsiness
be careful when driving a motor vehicle or operating machinery
If Pregnant Or Breast-Feeding:
If breast-feeding: not recommended
If pregnant: ask a health professional before use
Keep Out Of Reach Of Children.
In case of overdose, get medical help or contact a Poison Control Center right away at (1-800-222-1222).
Directions:
● may be taken with and without water
For Cetirizine hydrochloride chewable tablets 5 mg
adults and children 6 years and over | 1 to 2 tablets once daily depending upon severity of symptoms; do not take more than 2 tablets in 24 hours. |
adults 65 years and over | 1 tablet once a day; do not take more than 1 tablet in 24 hours. |
children under 6 years of age | ask a doctor |
consumers with liver or kidney disease | ask a doctor |
For Cetirizine hydrochloride chewable tablets 10 mg
adults and children 6 years and over | one 10 mg tablet once daily; do not take more than one 10 mg tablet in 24 hours. A 5 mg product may be appropriate for less severe symptoms. |
adults 65 years and over | ask a doctor |
children under 6 years of age | ask a doctor |
consumers with liver or kidney disease | ask a doctor |
Other information:
store between 20° to 25° C (68° to 77° F)
Phenylketonurics: Contains 1.68 mg Phenylalanine (a component of Aspartame) per 5 mg
Phenylketonurics: Contains 3.36 mg Phenylalanine (a component of Aspartame) per 10 mg
do not use if carton is opened or if blister unit is broken.
see bottom panel for lot number and expiration date.
Inactive Ingredients:
acesulfame potassium, artificial and natural flavors, aspartame, betadex, colloidal silicon dioxide, croscarmellose sodium, dl-alpha-tocopherol, ethyl cellulose, FD&C yellow # 6 aluminum lake, fumaric acid, hypromellose, magnesium stearate, mannitol, maltodextrin, microcrystalline cellulose and talc.
Questions? Call 1-800-313-4623
Manufactured by:
Jubilant Generics Ltd.
Roorkee-247661, India
Marketed by:
Jubilant Cadista Pharmaceuticals Inc.
Salisbury, MD 21801, USA
Revised : November / 2014
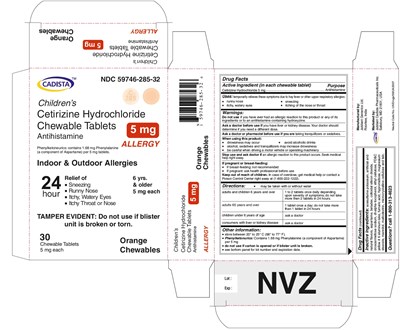
Package Label.Principal Display Panel
CADISTA NDC 59746-285-32
Children's
Cetirizine Hydrochloride Chewable Tablets 5 mg
Antihistamine
ALLERGY
Phenylketonurics: contains 1.68 mg Phenylalanine (a component of Aspartame) per 5 mg tablets.
Indoor & Outdoor Allergies
24 hour Relief of
● Sneezing
● Running Nose
● Itchy,Watery Eyes
● Itchy Throat or Nose
TAMPER EVIDENT : Do not use if blister unit is broken or torn.
6 yrs. & older
5 mg each
30 Chewable Tablets Orange Chewables
5 mg each
CADISTA NDC 59746-286-32
Children's
Cetirizine Hydrochloride Chewable Tablets 10 mg
Antihistamine
ALLERGY
Phenylketonurics: contains 3.36 mg Phenylalanine ( a component of Aspartame) per 10 mg tablets.
Indoor & Outdoor Allergies
24 hour Relief of
● Sneezing
● Running Nose
● Itchy,Watery Eyes
● Itchy Throat or Nose
TAMPER EVIDENT : Do not use if blister unit is broken or torn.
6 yrs. & older
10 mg each
30 Chewable Tablets Orange Chewables
10 mg each
* Please review the disclaimer below.