FDA Label for Antiseptic
View Indications, Usage & Precautions
Antiseptic Product Label
The following document was submitted to the FDA by the labeler of this product Safetec Of America. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Other
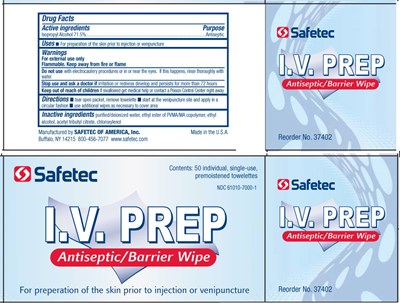
Drug Facts
Active Ingredients
Isopropyl Alcohol 71.5%
Purpose
Antiseptic
Uses
- For preparation of the skin prior to injection or venipuncture
Warnings
For external use only
Flammable. Keep away from fire or flame
Do not use with electrocautery procedures or in or near the eyes. If this happens, rinse thoroughly with water.
Otc - Stop Use
Stop use and ask doctor if irritation or redness develop and persists for more than 72 hours
Otc - Keep Out Of Reach Of Children
Keep out of reach of children If swallowed get medical help or contact a Poison Control Center right away
Directions
- start at the venipuncture site and apply in a circular fashion
- use additional wipes as necessary to cover area
Inactive Ingredients
purified/deionized water, ethyl ester of PVMA/MA copolymer, ethyl alcohol, acetyl tributyl citrate, chloroxylenol
Principal Display Panel – Packet Label
Safetec
I.V. Prep
Antiseptic/Barrier Wipe
For preparation of the skin prior to injection or venipuncture
Contents: 1 single-use, premoistened towelette
Manufactured by SAFETEC OF AMERICA, Inc.
Buffalo, NY 14215 800-456-7077 www.safetec.com
Principal Display Panel – Box
Safetec
Contents: 50 individual, single-use, premoistened towelettes
NDC 61010-7000-1
I.V. Prep
Antiseptic/Barrier Wipe
For preparation of the skin prior to injection or venipuncture
* Please review the disclaimer below.