Product Images Letairis
View Photos of Packaging, Labels & Appearance
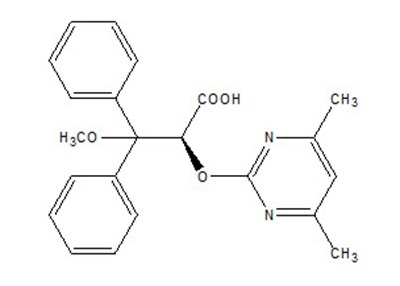
- Chemical Structure - letairis 01
- Figure 2 - letairis 02
- Figure 3 - letairis 03
- Figure 4 - letairis 04
- Figure 5 - letairis 05
- Figure 6 - letairis 06
- Figure 7 - letairis 07
- Figure 8 - letairis 08
- Figure 9 - letairis 09
- Chart - letairis 10
- PRINCIPAL DISPLAY PANEL - 5 mg Tablet Carton - letairis 11
- PRINCIPAL DISPLAY PANEL - 10 mg Tablet Carton - letairis 12
Product Label Images
The following 12 images provide visual information about the product associated with Letairis NDC 61958-0802 by Gilead Sciences, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 2 - letairis 02

This is a drug interaction table that shows the effects of various drugs on Ambrisentan. It provides recommendations for different drugs with regards to the dose adjustments for Ambrisentan. It includes Cyclosporine, Mycophenolate Mofetil, Ketoconazole, Omeprazole, Rifampin, Ritonavir, Sildenafil, Tacrolimus, Tadalafil, and Warfarin. The table also shows the fold change and 90% CI for Ambrisentan alone and in combination with different drugs. The recommendation for most drugs is no dose adjustment, except for Ambrisentan which is suggested to be limited to 5 mg once daily.*
Figure 3 - letairis 03

This text contains a list of drugs that interact with each other, along with their pharmacokinetics parameters such as Cmax and AUC. There is a table showing the fold change and 90% confidence interval. The recommendation is to not adjust the dose for all of the drugs listed.*
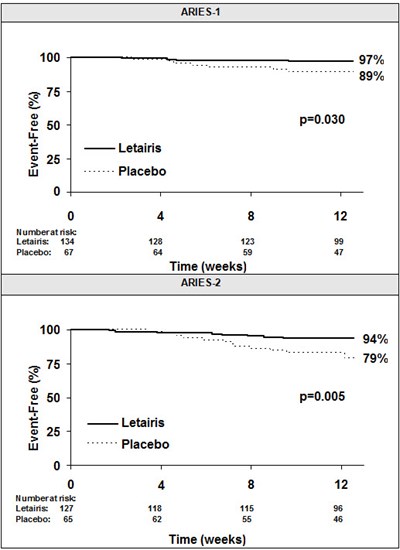
Figure 4 - letairis 04

This appears to be a chart or a graph representing different dosages of a medication, possibly placebo, and their effects over a 12-week period. It appears to be labeled as "ARIES-1" and "ARIES-2". It is not clear what the medication is, what condition it is treating, or what the specific effects are.*
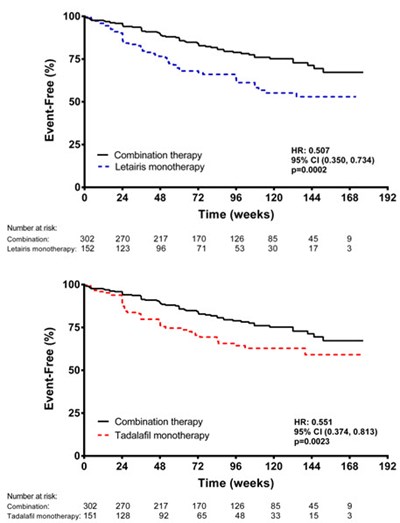
Figure 7 - letairis 07

This appears to be a table presenting Hazard Ratios and Confidence Intervals for different clinical events for a set of patients. The events include a primary endpoint, death, hospitalization, short term, and long term clinical response. The table also provides the number of patients with events, hazard ratios, 95% confidence intervals, and p-values. There are two types of treatments being compared - Combination vs. Letairis monotherapy and Combination vs. tadalafil monotherapy.*
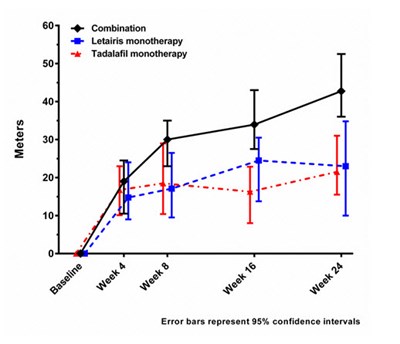
Figure 8 - letairis 08

This appears to be a table with various statistics related to the combination of treatments and monotherapy for different functional classes for patients with PAH and other conditions. It includes hazard ratios with confidence intervals for different regions and age groups. Further information on what the treatments are, the purpose of the statistics, or the study is not available.*
Chart - letairis 10

This text seems to be a list of different contraception methods, including the standard intrauterine device, intrauterine system, progesterone implant, and various hormonal contraceptives like the pill, transdermal patch, vaginal ring, and progesterone injection. It also includes barrier methods like male condoms, diaphragms with spermicide, and cervical caps with spermicide. Additionally, it suggests combining certain methods for added protection, such as using a male condom with a diaphragm or cervical cap with spermicide, and mentions the option of a partner's vasectomy.*
PRINCIPAL DISPLAY PANEL - 10 mg Tablet Carton - letairis 12

Letairis® is a medication available in the form of tablets containing 10mg of ambrisentan. The NDC number for Letairis is 61955-08023. The provided packaging has 10 tablets. There is a note asking the dispenser to provide a copy of the enclosed caution to every patient. Rest of the text is not readable.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.