Product Images Fluticasone Propionate And Salmeterol Hfa
View Photos of Packaging, Labels & Appearance
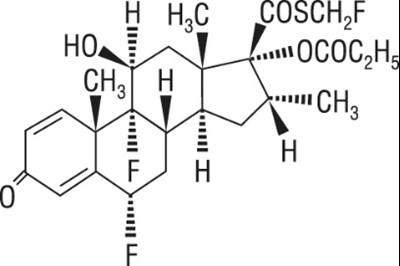
- Fluticasone chemical structure - fp sal hfa spl graphic 01
- Salmeterol chemical structure - fp sal hfa spl graphic 02
- Figure 1. Mean Percent Change from Baseline in FEV1 in Subjects Previously Treated with Either Beta2-agonists (Albuterol or Salmeterol) or Inhaled Corticosteroids (Trial 1) - fp sal hfa spl graphic 03
- Figure 2. Mean Percent Change from Baseline in Morning Peak Expiratory Flow in Subjects Previously Treated with Inhaled Corticosteroids (Trial 4) - fp sal hfa spl image 04
- Figure 3. Percent Change in Serial 12-Hour FEV1 in Subjects Previously Using Either Beta2-agonists (Albuterol or Salmeterol) or Inhaled Corticosteroids (Trial 1) - fp sal hfa spl image 05
- Figure 4. Percent Change in Serial 12-Hour FEV1 in Subjects Previously Using Either Beta2-agonists (Albuterol or Salmeterol) or Inhaled Corticosteroids (Trial 1) - fp sal hfa spl image 06
- Figure A - fp sal hfa spl image 07
- Figure B - fp sal hfa spl image 08
- Figure C - fp sal hfa spl image 09
- Figure D - fp sal hfa spl image 10
- Figure E - fp sal hfa spl image 11
- Figure F - fp sal hfa spl image 12
- Figure G - fp sal hfa spl image 13
- Figure H - fp sal hfa spl image 14
- Figure I - fp sal hfa spl image 15
- Figure J - fp sal hfa spl image 16
- Fluticasone Propionate and Salmeterol HFA 45 mcg-21 mcg 120 dose carton - fp sal hfa spl image 17
- Fluticasone Propionate and Salmeterol HFA 115 mcg-21 mcg 120 dose carton - fp sal hfa spl image 18
- Fluticasone Propionate and Salmeterol HFA 230 mcg-21 mcg 120 dose carton - fp sal hfa spl image 19
Product Label Images
The following 19 images provide visual information about the product associated with Fluticasone Propionate And Salmeterol Hfa NDC 66993-087 by Prasco Laboratories, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1. Mean Percent Change from Baseline in FEV1 in Subjects Previously Treated with Either Beta2-agonists (Albuterol or Salmeterol) or Inhaled Corticosteroids (Trial 1) - fp sal hfa spl graphic 03
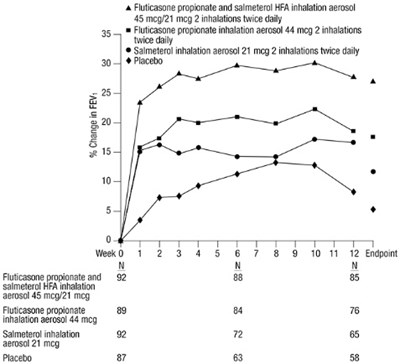
Figure 2. Mean Percent Change from Baseline in Morning Peak Expiratory Flow in Subjects Previously Treated with Inhaled Corticosteroids (Trial 4) - fp sal hfa spl image 04
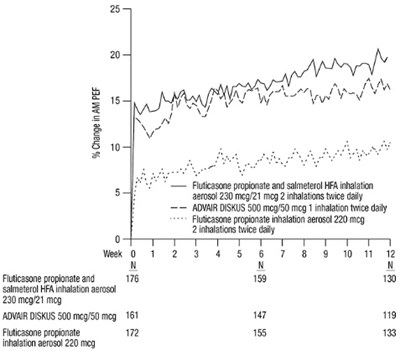
Figure 3. Percent Change in Serial 12-Hour FEV1 in Subjects Previously Using Either Beta2-agonists (Albuterol or Salmeterol) or Inhaled Corticosteroids (Trial 1) - fp sal hfa spl image 05
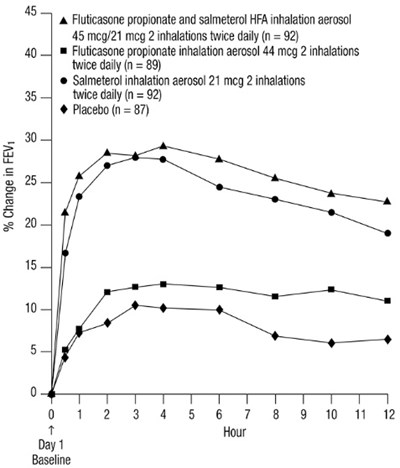
This appears to be a chart or table with medication dosages and a placebo. It includes information on the change in FEVy (likely a measure of lung function) over time for each treatment, as well as the number of participants in each group. Without more context, it is difficult to provide a more detailed description or interpretation of the data.*
Figure 4. Percent Change in Serial 12-Hour FEV1 in Subjects Previously Using Either Beta2-agonists (Albuterol or Salmeterol) or Inhaled Corticosteroids (Trial 1) - fp sal hfa spl image 06
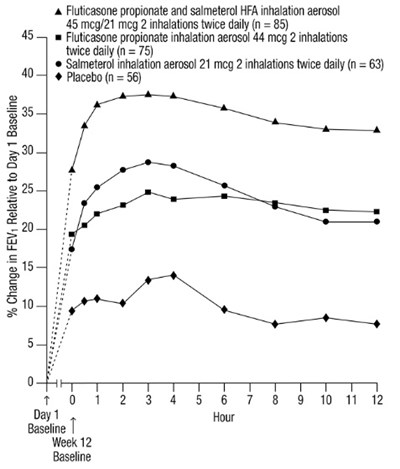
This text describes a study involving the use of two inhalers containing Fluticasone propionate and salmeterol HFA, as well as placebos. The study involved a total of 85 participants in the first group, who used the Fluticasone propionate and salmeterol inhaler, 75 participants in the second group, who used only the Fluticasone propionate inhaler, 63 participants in the third group, who used only the Salmeterol inhaler, and 56 participants in the placebo group. The study evaluated the change in FEV relative to the baseline on day 1, over a period of 12 weeks. However, the values of FEV changes are not provided in the text.*
Figure A - fp sal hfa spl image 07
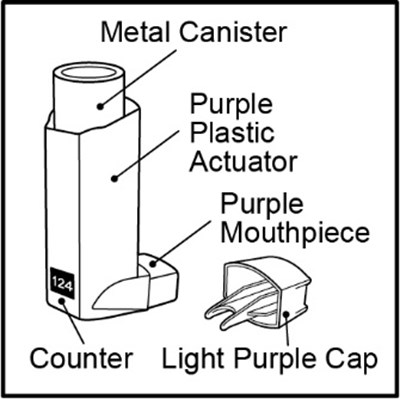
This is a metal canister with a purple plastic actuator and mouthpiece. It also has a counter light purple cap.*
Figure E - fp sal hfa spl image 11
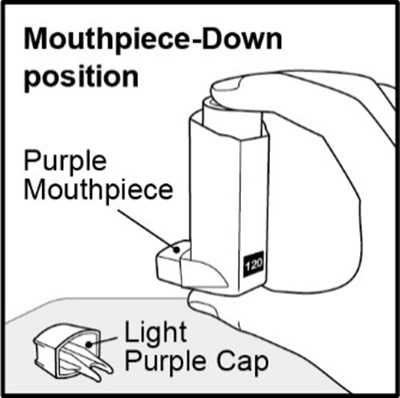
This is likely a product label or instruction manual for a device with a purple mouthpiece and cap. The label mentions a "Mouthpiece-Down position," but further context or information about the device is not available.*
Figure F - fp sal hfa spl image 12
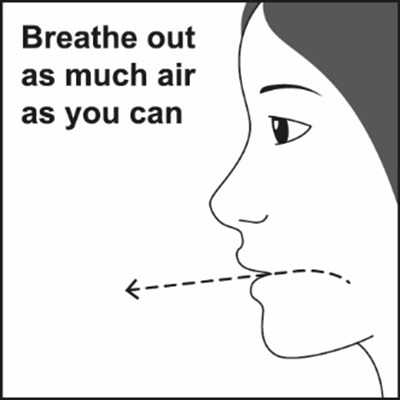
This is a simple instruction to help someone exhale as much air as possible.*
Fluticasone Propionate and Salmeterol HFA 45 mcg-21 mcg 120 dose carton - fp sal hfa spl image 17
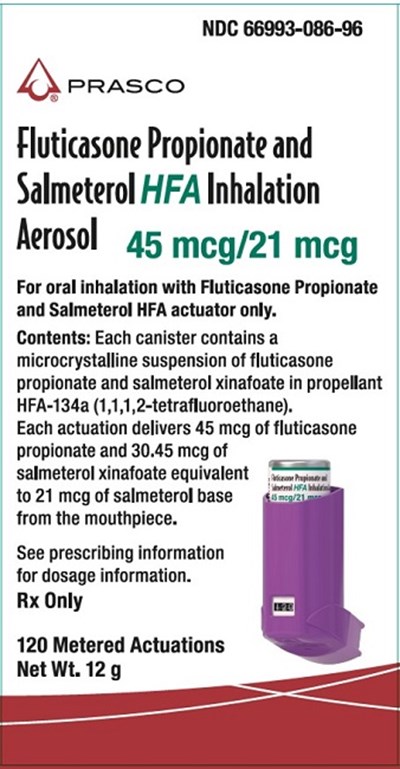
This is a description of a medication with the NDC number 66993-086-96, made by PRASCO. It is a Fluticasone Propionate and Salmeterol HFA Inhaler with Rerosol 45 mcg/21 meg. The medicine is for oral inhalation but only with Fluticasone Propionate and Salmeterol HFA actuator. The canister holds a microcrystalline suspension containing fluticasone propionate and salmeterol xinafoate in propellant HFA-134a. Each spray contains 45 mcg of fluticasone propionate and 30.45 mcg of salmeterol xinafoate equivalent et 1021 meg of salmeterol base gz from the mouthpiece. It has 120 metered actuations and a net weight of 12g. Dosage information can be found in the prescribing information. It is a prescription drug.*
Fluticasone Propionate and Salmeterol HFA 115 mcg-21 mcg 120 dose carton - fp sal hfa spl image 18

This text is a description of a medication named Fluticasone Propionate and Salmeterol HFA Inhalation. It contains the drug's name, the manufacturer's name (PRASCO), and the product's NDC code (66993-087-96). It specifies that the medication is for oral inhalation and comes in a canister that contains a microcrystalline suspension of Fluticasone Propionate and Salmeterol Xinafoate in propellant HFA-134a. The medication's dosage information can be found in the prescribing information. The medication is only available with a prescription. The product contains 120 metered actuations and weighs 12g.*
Fluticasone Propionate and Salmeterol HFA 230 mcg-21 mcg 120 dose carton - fp sal hfa spl image 19

This is the description for NDC 66993-088-96, a Fluticasone Propionate and Salmeterol HFA Inhalation aerosol for oral inhalation. It has 230 mcg of Fluticasone Propionate and 30.45 mcg of Salmeterol Xinafoate equivalent to 21 mcg of salmeterol base for each actuation. It comes in a canister with 120 metered actuations and is only for use with Fluticasone Propionate and Salmeterol HFA actuator. Prescription is required and dosage information is provided in the prescribing information.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.