Zafirlukast Tablet, Film Coated
Product Images NDC 68084-059
View Photos of Packaging, Regulatory Labels, and Product Appearance
Product Visual Gallery
This gallery contains 5 technical images submitted to the FDA as part of the official labeling for Zafirlukast (NDC 68084-059). Unlike standard consumer photos, these assets often include clinical data figures, molecular chemical structures, and official manufacturer packaging layouts.
As provided by American Health Packaging, these visuals offer a comprehensive scientific overview of the product's physical and chemical identity, aiding pharmacists and researchers in product verification and study.
20 mg Zafirlukast Tablets Carton (20 mg Zafirlukast Tablets Carton)

This is a description of Zafirlukast tablets, which come in a bottle of 30 tablets. The recommended dosage is specified in the package insert. The tablets should be stored at controlled room temperature and protected from light and moisture. The accompanying patient information sheet needs to be dispensed. The manufacturer is American Health Packaging, and the drug product is from Dr. Reddy's Laboratories Limited. The packaging includes the National Drug Code (NDC) number.*
8cc7d663 Fb49 459e E053 2995a90aeabd
Mean Beta2 (agonist use (puffs/day))
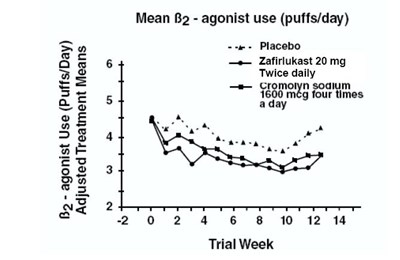
This text provides information on the average use of B2-agonist, in terms of puffs per day, in a clinical trial. The trial tests the effectiveness of different treatment strategies, including a placebo, a drug called Zafirlukast taken twice daily, and Gromolyn sodium taken four times a day at a dosage of 1600 mcg. The results are presented in a graph, which covers a 14-week period of the trial.*
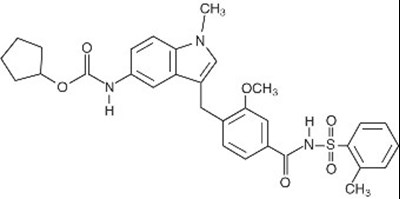
Zafirlukast Structural Formula (Image 02)
* These product label images have been analyzed using experimental machine learning. Please verify findings with the primary label text.