Product Images Abilify
View Photos of Packaging, Labels & Appearance
- Aripiprazole Chemical Structure - Abilify Chemical Structure
- Figure 1 - Abilify Figure 1
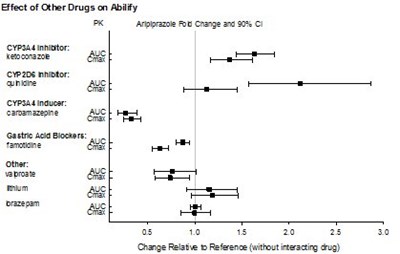
- Figure 2 - Abilify Figure 2
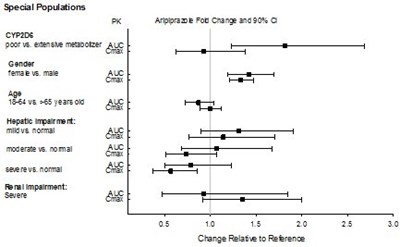
- Figure 3 - Abilify Figure 3
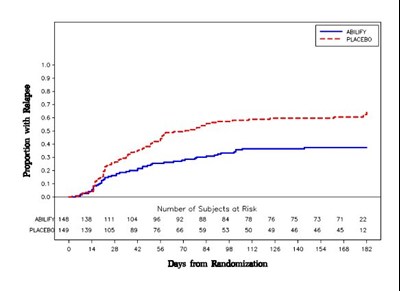
- Figure 4 - Abilify Figure 4
- Figure 5 - Abilify Figure 5
- Figure 6 - Abilify Figure 6
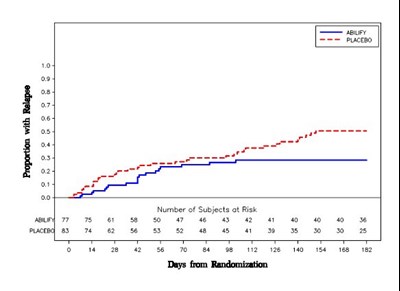
- Figure 7 - Abilify Figure 7
- Figure 8 - Abilify Figure 8
- Figure 9 - Abilify Figure 9
- Otsuka Logo - Abilify Otsuka Logo
- Label Image - lbl681515121
Product Label Images
The following 12 images provide visual information about the product associated with Abilify NDC 68151-5121 by Carilion Materials Management, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Otsuka Logo - Abilify Otsuka Logo

Oisuko is not a recognizable word, however, Otsuka America Pharmaceutical, Inc. is a pharmaceutical company based in the United States.*
Label Image - lbl681515121

ABILIFY (aripiprazole) is a medication manufactured by Otsuka in 2mg tablets. It is a prescription drug that should only be taken under the supervision of a licensed healthcare professional. The batch number is 10T:3778211 and the expiration date is 08/14. The PDM number is 5121, and the label reads "RX Only". The other characters, including "JZLIH", are not readable.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.