Product Images Minivelle
View Photos of Packaging, Labels & Appearance
- Estradiol USP - minivelle figure 1
- Figure D - minivelle figure 10
- Figures E and F - minivelle figure 11
- Figure G - minivelle figure 12
- trade carton 0.025mg - minivelle figure 13
- CARTON 0.0375mg - minivelle figure 14
- CARTON 0.05mg - minivelle figure 15
- CARTON 0.075mg - minivelle figure 16
- CARTON 0.1mg - minivelle figure 17
- Layers of MINIVELLE - minivelle figure 2
- Figure 1: Mean Baseline-Uncorrected Estradiol Serum Concentration-Time Profiles Following a Single Dose of MINIVELLE 0.1 mg per day (Treatment A), 0.05 mg per day (Treatment B), and 0.025 mg per day ( - minivelle figure 3
- Figure 2: Mean (SD) change from baseline in mean daily number of hot flushes for Vivelle 0.0375 mg versus Placebo in a 12 week trial. - minivelle figure 4
- Figure 3: Bone mineral density – AP Lumbar spine - minivelle figure 5
- Figure 4 Bone mineral density - Femoral neck - minivelle figure 6
- Figure A - minivelle figure 7
- Figure B - minivelle figure 8
- Figure C - minivelle figure 9
Product Label Images
The following 17 images provide visual information about the product associated with Minivelle NDC 68968-6610 by Noven Therapeutics, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
CARTON 0.075mg - minivelle figure 16

Minivelle is a transdermal system that contains 1.24 mg of estradiol USP, which delivers 0.075 mg/day. The system should be applied twice-weekly and contains an acrylic adhesive, silicone adhesive, and other inactive ingredients. It should be stored at 20°C to 25°C and should only be used transdermally. The package should be kept out of children's reach, and applied immediately after removing it from the pouch. Dosage and administration instructions can be found in the package insert. The manufacturer is Noven Pharmaceuticals, Inc. and the distributor is Noven Therapoutics, LLC, both located in Miami, Florida.*
CARTON 0.1mg - minivelle figure 17

This appears to be a product description for Minivelle, a transdermal system that delivers 0.1mg/day of estradiol USP. It comes in a package containing 8 systems and is applied twice-weekly. The product contains acrylic and silicone adhesive, oleyl alcohol, dipropylene glycol, among other inactive components. Package should be kept out of reach of children and stored at a controlled room temperature. The manufacturer is Noven Pharmaceuticals, Inc, and the distributor is Noven Therapoutics, LLC from Miami, Florida.*
Layers of MINIVELLE - minivelle figure 2

The product is an adhesive containing estradiol and comes with a protective liner.*
Figure 1: Mean Baseline-Uncorrected Estradiol Serum Concentration-Time Profiles Following a Single Dose of MINIVELLE 0.1 mg per day (Treatment A), 0.05 mg per day (Treatment B), and 0.025 mg per day ( - minivelle figure 3
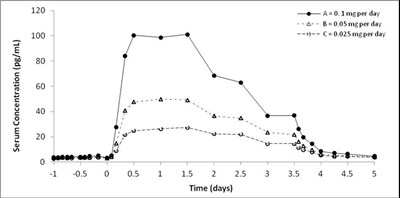
Figure 2: Mean (SD) change from baseline in mean daily number of hot flushes for Vivelle 0.0375 mg versus Placebo in a 12 week trial. - minivelle figure 4
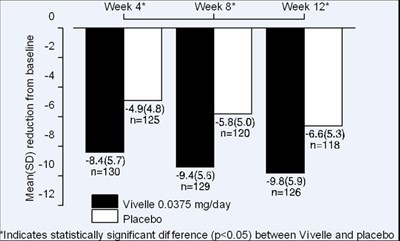
The text describes statistical data of a study conducted over 12 weeks, with measurements taken at weeks 4, 8, and 12. The data includes numeric values for different variables and a comparison between a treatment called Vivelle and a placebo. The last sentence suggests that there was a statistically significant difference between the two treatments.*
Figure 3: Bone mineral density – AP Lumbar spine - minivelle figure 5

This is a table or graph showing the % change from baseline for different treatments at different time points (Week 2, Week 26, Week 78, and Week 104). The treatments compared include Vivelle 0.1mg/day, Vivelle 0.025mg/day, Vivelle 0.05mg/day, Vivelle 0.0375mg/day, and Placebo.*
Figure 4 Bone mineral density - Femoral neck - minivelle figure 6
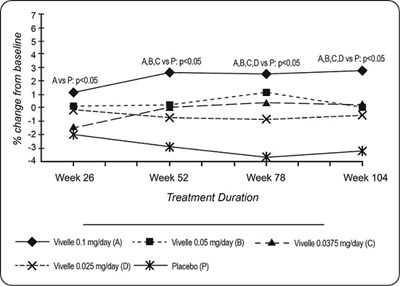
This text appears to be a table showing the percentage change from baseline for various treatments over the course of several weeks. The treatments are labeled A, B, C, D, and P. There are statistical values listed with some of the treatments, including p-values. The full extent of this data set is not available and any further interpretation of the data is not possible.*
Figure A - minivelle figure 7

This text is a list of three words "PIPPI", "Calendar", and "Pouch containing patch". It seems to describe a product or products, likely related to organization or storage.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.








