Product Images Netspot
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 12 images provide visual information about the product associated with Netspot NDC 69488-001 by Advanced Accelerator Applications Usa, Inc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
carton - carton

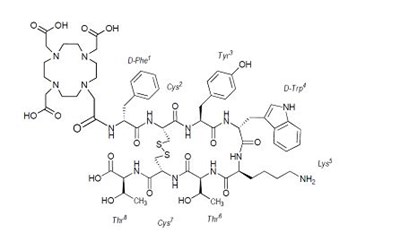
The text describes a kit designed for preparing an injection of Ga 68 dotatate for intravenous use.*
fig1 - fig1

This is a procedure for the production of 68Ga and 58Ga generators, including reaction buffers, eluate, and dotatate. The process involves heating at 95°C for seven minutes followed by cooling at room temperature for ten minutes, with activity measurement and visual inspection determining whether or not to pass. The final step involves a release of Ga 68 dotatate.*
fig2 - fig2
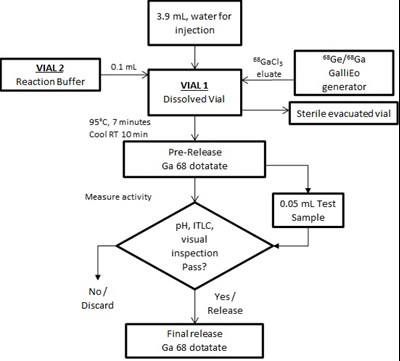
This appears to be a protocol or set of instructions for measuring the activity of a substance called "Ga 68 dotatate" using various chemicals and equipment such as a generator, dissolved vials, and an injection. The text includes specific temperatures and instructions for measuring pH and visual inspection to determine if the substance passes or fails. Overall, it seems to be a process for quality control in the manufacturing or production of this substance.*
reconstitution - reconstitution
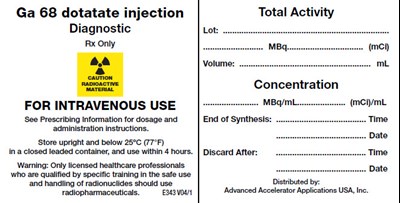
This is a description of Ga 68 dotatate injection, a diagnostic radiopharmaceutical product meant for intravenous use. It is only intended for use by licensed healthcare professionals who have undergone specific training in the safe handling of radionuclides. The prescribing information should be consulted for dose and administration instructions. The product must be stored upright at below 25°C and used within four hours of opening the container. The product details include lot number, total activity, volume, concentration, and end of synthesis time, and discard after time and date. The product must be discarded after this time. This product is distributed by Advanced Accelerator Applications USA, Inc.*
syringe - syringe

NETSPOT® is a kit that provides a syringe for the preparation of Ga 68 dotatate Injection. It is important to refer to the prescribing information before using this injection. The reconstituted injection should be used within 4 hours. The text also includes a code, E331 V0472.*
vial1 01

This is a medication description for a single dose vial containing 40 micrograms of dotatate. The vial is meant for intravenous use only and any unused portion should be discarded. The medication is stored in a "Reaction vial" with lyophilized powder. The lot number and expiration date are also included as important information.*
vial1 02

This text appears to be a medication label with information on a specific drug. The drug is identified as NDC 69488-001-04 and is only available with a prescription. It comes in a single-dose vial and any unused portion should be discarded. The drug requires reconstitution with Ga 68 chloride and pH adjustment with a Reaction Buffer before use. It is also noted that the drug is sterile. No specific dosage information is provided and it is recommended that the prescribing information be consulted for further details.*
vial1 03

The text provides storage instructions for NETSPOT®. It should be stored at a temperature below 25°C (77°F) and not frozen. After reconstitution, it should be stored upright in a closed container and used within 4 hours.*
vial2 04

This is a label of a pharmaceutical product with NDC code 69488-001-01. It is a single-dose vial for Rx use only, and any unused portion of the product should be discarded. The product is intended for adjusting the pH of a radiopharmaceutical, specifically Ga-68 dotatate injection. The label provides instructions for pH adjustment of a radiolabeled product called Netspot using a volume of 1 mL in a 10 mL vial. The text contains a few optical character recognition errors but is mostly readable.*
vial2 05

NETSPOT® is a reaction buffer used for preparing Gallium-68 dotatate injection. It consists of 60 mg of formic acid, 56.5 mg of sodium hydroxide, and water for injection. The buffer is sterile but not intended for direct administration.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.