Product Images Nerlynx
View Photos of Packaging, Labels & Appearance
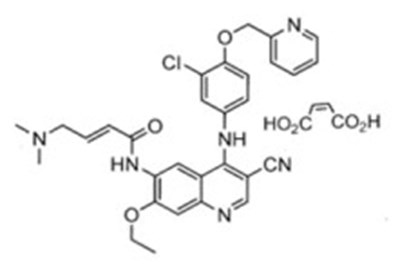
- Structural Formula - ner00 0019 01
- Figure 1 - ner00 0019 02
- Figure - ner00 0019 03
- Figure - ner00 0019 04
- Principal Display Panel - Nerlynx 180 Tablets Carton Label - ner00 0019 05
- Principal Display Panel - Nerlynx 180 Tablets Bottle Label - ner00 0019 06
- Principal Display Panel - Nerlynx 126 Tablets Carton Label - ner00 0019 07
- Principal Display Panel - Nerlynx 126 Tablets Bottle Label - ner00 0019 08
- Principal Display Panel - Nerlynx 133 Tablets Carton Label - ner00 0019 09
- Principal Display Panel - Nerlynx 133 Tablets Bottle Label - ner00 0019 10
Product Label Images
The following 10 images provide visual information about the product associated with Nerlynx NDC 70437-240 by Puma Biotechnology, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1 - ner00 0019 02
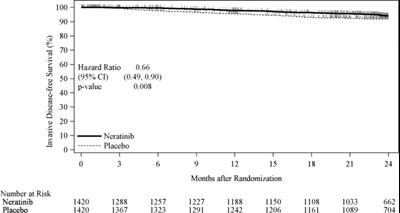
This text provides data on invasive disease-free survival in patients who were either treated with Neratinib or a placebo. The data includes the number of patients at risk, hazard ratio, and percentage of survival over time in months.*
Figure - ner00 0019 03
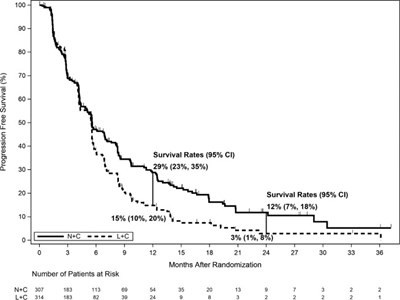
The text represents a graphical representation of Progression Free Survival and Survival Rates for a certain period of time after randomization. It includes the percentage values and Number of Patients at Risk for different time intervals shown on the X-axis. The Survival rates with a 95% confidence interval are also provided for different intervals. The text suggests the study might be related to cancer treatment, although the exact medical condition is not clear.*
Figure - ner00 0019 04
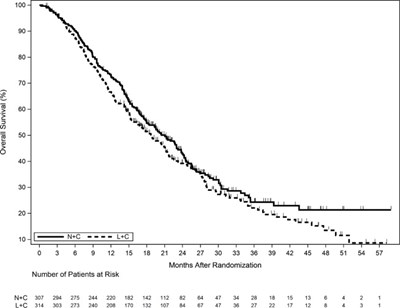
This is a graph showing the overall survival of patients over time after randomization. The x-axis represents months and the y-axis represents the percentage of survival. The graph includes the number of patients at risk during each time interval. No further information can be extracted due to the errors in the text.*
Principal Display Panel - Nerlynx 180 Tablets Carton Label - ner00 0019 05
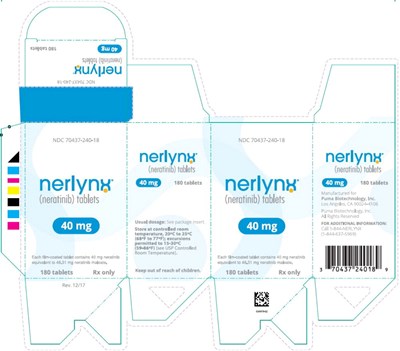
This is a description for a medication with the brand name Nerlynix. It comes in the form of tablets and is only available with a prescription. The usual dosage is not mentioned. The medicine should be stored between 68°F to 77°F, with excursions permitted to 15-30°C (59-86°F). The National Drug Code (NDC) for this medication is 70437-240-18. The tablets are produced by Puma Biotechnology, Inc. and the company is located in Los Angeles, CA, USA.*
Principal Display Panel - Nerlynx 180 Tablets Bottle Label - ner00 0019 06

This text describes information related to the drug NDC 70437-240-18. It provides details about the manufacturer and dosage of the drug. The tablets are film-coated and contain neratinib, equivalent to 4831 mg of neratinib maleate. The text also suggests to refer to the package insert for instructions on the usual dosage. It also mentions that the drug is an Rx only medication and should be kept out of the reach of children.*
Principal Display Panel - Nerlynx 126 Tablets Carton Label - ner00 0019 07
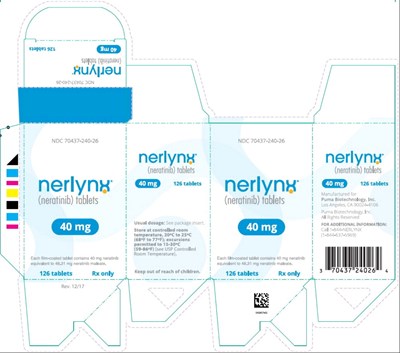
Product Name: Nerlyn Tablets Quantity: 126 tablets Active ingredient: Neratinib Dosage: Not specified Storage: Store at room temperature Caution: Keep out of reach of children. NDC code: 70437-240-26*
Principal Display Panel - Nerlynx 126 Tablets Bottle Label - ner00 0019 08

This is a description of a medication marketed under the NDC 70437-240-26 code known as "nerlyni". It contains neratinib and comes in a package of 126 tablets. It is a prescription-only medication and must be kept at a controlled room temperature of 20°C to 25°C, with some allowable excursions. The manufacturer is Puma Biotechnology, Inc. and the recommended dosage information can be found in the package insert. Each tablet has a protective film layer and contains a code "€(01)10370". The label warns to keep the tablets away from children.*
Principal Display Panel - Nerlynx 133 Tablets Carton Label - ner00 0019 09

This is a description of a medication called "nerlyny" or "neratinib" tablets. It comes in a package with 133 tablets and the usual dosage information can be found in the package insert. The medication should be stored at a controlled room temperature between 20°C to 25°C (63°F to 77°F) and should be kept out of reach of children. The tablets are manufactured for Puma Biotechnology, Inc in Los Angeles, CA. For additional information, call 1-844-637-5969.*
Principal Display Panel - Nerlynx 133 Tablets Bottle Label - ner00 0019 10

This is a description of a medication named "Nerlyn" (generic name: neratinib) which comes in tablet form. The National Drug Code (NDC) for this medication is 70437-240-33, and it should be stored at a controlled room temperature between 20°C to 25°C (68°F to 77°F), with excursions permitted to 15-30°C (59-86°F) as per the USP Controlled Room Temperature. Each tablet contains 40 mg of neratinib, which is equivalent to 4831 mg of neratinib maleate. This medication is prescription-only and has a recommended dosage that can be found in the package insert. This medication is manufactured for Puma Biotechnology, Inc. in Los Angeles, CA.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.