Product Images Fulvestrant
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 18 images provide visual information about the product associated with Fulvestrant NDC 70700-284 by Xiromed, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
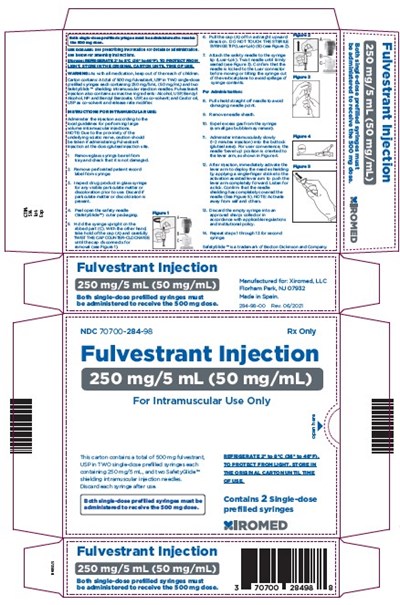
container label

This appears to be a description of a pharmaceutical product called "Fulvestrant Injection" indicated for intramuscular use only. The injection comes in a single-dose prefilled syringe, and two syringes should be administered to receive the recommended dose of 500 mg. The NDC number for this product is 70700-284-98, and it is manufactured for Xiromed, LLC in Florham Park, NJ. There are also measurements listed in millimeters, but it is not clear what they refer to.*
Figure-12 - fulvestrant inj fig 12

The text represents a Survival Probability Graph comparing the effectiveness of two treatments - FULVESTRANT plus Abemaciclib and FULVESTRANT plus Placebo on a sample size of 446 and 223 respectively. However, due to 100 censored observations, the time scale and the data are not fully available to generate a full interpretation of the survival rate.*
Figure-6 - fulvestrant inj fig 6

The text describes a table showing the number of patients at risk over time (in months) for two different doses of a medication called Fulvestrant (250mg and 500mg). The table seems to show the number of patients who completed the study without experiencing an event, for each time interval.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.