Product Images Duloxetine Delayed-release
View Photos of Packaging, Labels & Appearance
- Label - 70934 729 30 Master Bottle Label Approval Rev A 08 20
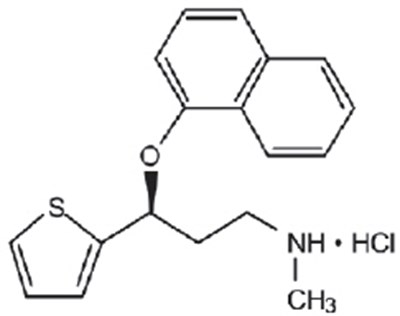
- Chemical Structure - duloxetine 01
- Figure 1 - duloxetine 02
- Figure 2 - duloxetine 03
- Figure 3 - duloxetine 04
- Figure 4 - duloxetine 05
- Figure 5 - duloxetine 06
- Figure 6 - duloxetine 07
- Figure 8 - duloxetine 08
- Figure 9 - duloxetine 09
- Figure 10 - duloxetine 10
Product Label Images
The following 11 images provide visual information about the product associated with Duloxetine Delayed-release NDC 70934-729 by Denton Pharma, Inc. Dba Northwind Pharmaceuticals, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Label - 70934 729 30 Master Bottle Label Approval Rev A 08 20

This is a medication called Duloxetine, which is in the form of delayed-release capsules. It comes in a bottle containing 30 capsules, with each capsule weighing 337 mg, and the bottle has an NDC code of 70934-729-30. The rest of the text is not readable.*
Figure 1 - duloxetine 02

This is a graph showing the proportion of patients with relapse based on the treatment they received (either a Placebo or Duloxstine delayed-release capsules) over time (measured in days). The graph displays percentages ranging from 0% to 10% on the Y-axis and time ranging from 0 to 250 days on the X-axis.*
Figure 2 - duloxetine 03
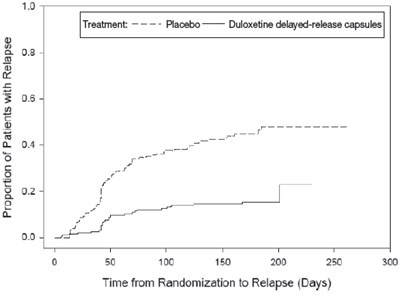
This is a graph showing the proportion of patients with relapse for two treatments: placebo and Duloxetine delayed-release capsules. The x-axis represents time from randomization to relapse (in days), and the y-axis represents the proportion of patients with relapse. It appears that the group treated with Duloxetine delayed-release capsules had a lower proportion of patients with relapse over time compared to the placebo group.*
Figure 8 - duloxetine 08

This text appears to be a statistical representation of the percentage of patients who showed improvement in pain after being treated with either a placebo or DUL 60/120 mg daily. The percentages are shown in numbers and divided into different categories. Additionally, there is a graph illustrating the percentage improvement in pain from baseline. The text seems incomplete and does not provide further context or details for the statistical data.*
Figure 9 - duloxetine 09

The text shows a chart or graph displaying the percentage of patients who experienced an improvement with a certain treatment, possibly related to pain relief. The treatment being evaluated is likely a medication, as there are two groups compared: a placebo and a 60mg once-daily dose of DUL (could be an abbreviation for a medication name). It also shows a scale ranging from 50 to 100 along the horizontal axis that indicates the percentage of improvement in pain from the baseline (BOCF).*
Figure 10 - duloxetine 10

This is a graph showing the percentage improvement in pain from baseline for patients taking either a placebo or Duloxetine (DUL) at either 60mg or 120mg doses, once daily. The graph ranges from 0 to 100 and shows the percentage of patients who have improved their pain levels. There is no further information available.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.