Product Images Repatha
View Photos of Packaging, Labels & Appearance
- repatha amgenusa 01
- GRH0552 GRAPH MISC NA v1.0 - repatha amgenusa 02
- GRH0553 GRAPH MISC NA v1.0 - repatha amgenusa 03
- GRH0554 GRAPH MISC NA v1.0 - repatha amgenusa 04
- GRH0434 GRAPH MISC NA v1.0 - repatha amgenusa 05
- GRH0555 GRAPH MISC NA v2.0 - repatha amgenusa 06
- repatha amgenusa 07
- Prefilled Cartridge - repatha amgenusa 08
- On Body Infusor - repatha amgenusa 09
- Open the on body infusor by swinging the cartridge door to the right. Then, leave the door open. Do not close the cartridge door before the cartridge is loaded. - repatha amgenusa 10
- Inspect the cartridge. - repatha amgenusa 11
- Clean the cartridge bottom. - repatha amgenusa 12
- Loadcartridgestraight - repatha amgenusa 13
- Press downfirmly - repatha amgenusa 14
- Swing the door to the left. Then, squeeze firmly until it snaps shut. Apply enough pressure when closing the door and make sure there is a “snap” before going to the next step. - repatha amgenusa 15
- Peel away both green pull tabs to show the adhesive. The on body infusor is on when the blue status light flashes. - repatha amgenusa 16
- Stomach area placement - repatha amgenusa 17
- Thigh placement - repatha amgenusa 18
- When the blue light flashes, the on body infusor is ready. Keep the stretch (stomach area method only). Hold the loaded on body infusor with the blue light visible, and place it on your skin. You may hear beeps. - repatha amgenusa 19
- What do I do if the loaded on body infusor status light continuously flashes red and I hear beeps - repatha amgenusa 20
- Flashing warning light - repatha amgenusa 21
- beep-beep-beep-beep- beep - repatha amgenusa 22
- repatha amgenusa 23
- Do not re-use - repatha amgenusa 24
- Serial number - repatha amgenusa 25
- Type BF Applied Part - repatha amgenusa 26
- Do not use if packaging is damaged - repatha amgenusa 27
- On-Body Infusor containing420 mg/ 3.5 mL (120 mg/mL) - repatha amgenusa 28
- Sterilized using ethylene oxide - repatha amgenusa 29
- Wait at least 30 minutes for the autoinjector to reach room temperature before injecting - repatha amgenusa 30
- Inspect the Repatha SureClick autoinjector. - repatha amgenusa 31
- New autoinjector, Alcohol wipes, Cotton ball or gauze pad, Adhesive bandage, Sharps disposal container (see Step 4: Finish) - repatha amgenusa 32
- Prepare and clean your injection site. - repatha amgenusa 33
- Pull the orange cap off only when you are ready to inject. - repatha amgenusa 34
- Do not leave the orange cap off for more than five minutes. This can dry out the medicine. - repatha amgenusa 35
- Thigh:Stretch method - repatha amgenusa 36
- Stomach or upper arm:Pinch method - repatha amgenusa 37
- Hold the stretch or pinched skin. With the orange cap off, place autoinjector on the skin at 90 degrees. Do not touch the gray start button yet. - repatha amgenusa 38
- Firmly push down the autoinjector onto the skin until it stops moving. - repatha amgenusa 39
- Before use, guide to parts - repatha amgenusa 40
- Guide to parts- after use - repatha amgenusa 41
- Remove the Repatha prefilled syringe carton from the refrigerator and wait 30 minutes. - repatha amgenusa 42
- 1 Rapatha prefilled syringe - repatha amgenusa 43
- Choose your injection site. - repatha amgenusa 44
- Clean your injection site with an alcohol wipe. Let your skin dry before injecting - repatha amgenusa 45
- Remove prefilled syringe from tray. - repatha amgenusa 46
- Check the medicine and syringe. - repatha amgenusa 47
- Carefully pull the gray needle cap straight out and away from your body. - repatha amgenusa 48
- Place the cap in the sharpsdisposal container right away. - repatha amgenusa 49
- PRINCIPAL DISPLAY PANEL1 x 1 mL Prefilled SyringeNDC 72511-750-01Amgen®Repatha® (evolocumab)Injection140 mg/mL140 mg/mLPrefilled SyringeFor Subcutaneous Use OnlyStore at 2°C to 8°C (36°F to 46°F). Do Not Freeze or Shake.Store in Carton to Protect from Light.(see side panel for additional storage information)Sterile Solution – No PreservativeSingle UseRx OnlyCAUTION, See package insert for full prescribing information and Instructions for UseThis Product Contains Dry Natural Rubber.Keep out of the sight and reach of children - repatha amgenusa 50
- PRINCIPAL DISPLAY PANEL1 x 3.5 mL Prefilled Cartridge1 On-Body InfusorNDC 72511-770-01Amgen®Repatha® Pushtronex® system(evolocumab)On-Body Infusor and Prefilled Cartridge420 mg/3.5 mL420 mg/3.5 mLFor Subcutaneous Use OnlySingle-Use OnlySterile Solution – No PreservativeStore refrigerated at 2°C to 8°C (36°F to 46°F). Do Not Freeze or Shake.Store in Carton to Protect from Light.(see side panel for additional storage information)Keep out of the sight and reach of children.Refer to Instructions for UseDo Not Use if Package is DamagedOn-Body Infusor Sterilized Using Ethylene OxideKeep DryDo not re-useType BF Applied PartRelative HumidityRange is 15% to 85%Rx Only - repatha amgenusa 51
- repatha amgenusa 52
- repatha amgenusa 53
- repatha amgenusa 54
- repatha amgenusa 55
- repatha amgenusa 56
- repatha amgenusa 57
- repatha amgenusa 58
- repatha amgenusa 59
- repatha amgenusa 60
- repatha amgenusa 61
- repatha amgenusa 62
- repatha amgenusa 63
- repatha amgenusa 64
- repatha amgenusa 65
- repatha amgenusa 66
- repatha amgenusa 67
- repatha amgenusa 68
- repatha amgenusa 69
- repatha amgenusa 70
- repatha amgenusa 71
- repatha amgenusa 72
- repatha amgenusa 73
- repatha amgenusa 74
- repatha amgenusa 75
- repatha amgenusa 76
- repatha amgenusa 77
- repatha amgenusa 78
- repatha amgenusa 79
- repatha amgenusa 80
- repatha amgenusa 81
- repatha amgenusa 82
- repatha amgenusa 83
- repatha amgenusa 84
- repatha amgenusa 85
- repatha amgenusa 86
Product Label Images
The following 86 images provide visual information about the product associated with Repatha NDC 72511-770 by Amgen Usa Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
repatha amgenusa 01
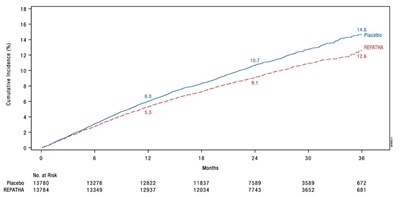
This appears to be a description of a clinical trial with two groups: Placebo and REPATHA. The data provided includes the number of people at risk over time for both groups, measured in months.*
GRH0554 GRAPH MISC NA v1.0 - repatha amgenusa 04

This is a graph that shows the percentage change from baseline in response to different treatments at different time points. The treatments compared are a placebo administered once a month and a medication called Repatha given at a dose of 420mg also administered once a month. The time points evaluated are at week 12, week 24, week 36, and week 52. The data was obtained from a study that included 302 individuals who received the placebo and 599 individuals who received Repatha.*
GRH0555 GRAPH MISC NA v2.0 - repatha amgenusa 06

This is a table that shows the number of subjects and their assigned treatments in a study. The study lasted for 12 weeks and the treatments were either placebo or EvoMab 420 mg. There were 53 subjects in the placebo group and 104 subjects in the EvoMab group.*
repatha amgenusa 07

This appears to be a partial product label or instructions for a medication delivery device. It mentions a "White plunger," a "Cartridge top," and a "Cartridge bottom" which may be parts of the device. It also references a label for a "Medicine Cartridge." Without more context, it is difficult to determine the exact nature or purpose of the device.*
Prefilled Cartridge - repatha amgenusa 08

The text describes the components of a device, presumably a medical injector. It includes a cartridge door with a warning not to close it without a cartridge, a skin adhesive light with a warning not to press it until ready to inject, and a medicine window along with pull tabs.*
On Body Infusor - repatha amgenusa 09
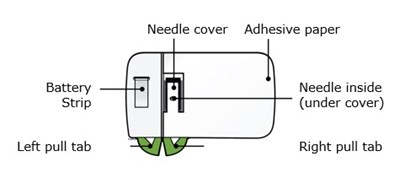
This is a description of a needle cover made out of adhesive paper. The cover encloses a battery needle and a strip is located underneath the cover. The cover has a left and right pull tab for easy removal.*
Open the on body infusor by swinging the cartridge door to the right. Then, leave the door open. Do not close the cartridge door before the cartridge is loaded. - repatha amgenusa 10
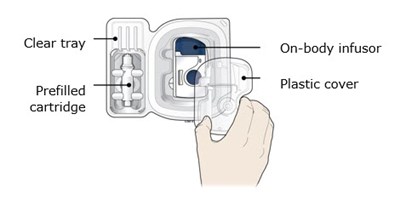
This appears to be a list of items related to a medical device. It includes a "clear tray", an "on-body infusor", a "prefilled T plastic cover", and a "cartridge". The purpose or function of the device is not clear from this text.*
Clean the cartridge bottom. - repatha amgenusa 12

This text appears to be a list of body parts: Upper arm, stomach area (abdomen), and thigh. It could be useful for someone who needs to identify or describe these body areas in a medical or fitness context.*
Swing the door to the left. Then, squeeze firmly until it snaps shut. Apply enough pressure when closing the door and make sure there is a “snap” before going to the next step. - repatha amgenusa 15

Peel away both green pull tabs to show the adhesive. The on body infusor is on when the blue status light flashes. - repatha amgenusa 16

This appears to be a set of instructions for using a product that involves loading a cartridge and injecting something within five minutes of doing so. The text advises pressing down firmly to ensure proper loading. However, without additional context or information, it is difficult to determine what type of product this is or what the injection is being used for.*
Thigh placement - repatha amgenusa 18

This text seems to be a list of three items: left and right pull tabs, and skin adhesive. The last line, "beep-beep-beep," is not clear what it refers to and may not be applicable to the list. Therefore, the list could be describing a product that includes left and right pull tabs and skin adhesive. However, without more context, it is difficult to determine what the product is or how these items are used.*
When the blue light flashes, the on body infusor is ready. Keep the stretch (stomach area method only). Hold the loaded on body infusor with the blue light visible, and place it on your skin. You may hear beeps. - repatha amgenusa 19
What do I do if the loaded on body infusor status light continuously flashes red and I hear beeps - repatha amgenusa 20

Wait at least 30 minutes for the autoinjector to reach room temperature before injecting - repatha amgenusa 30

New autoinjector, Alcohol wipes, Cotton ball or gauze pad, Adhesive bandage, Sharps disposal container (see Step 4: Finish) - repatha amgenusa 32

Do not leave the orange cap off for more than five minutes. This can dry out the medicine. - repatha amgenusa 35

Hold the stretch or pinched skin. With the orange cap off, place autoinjector on the skin at 90 degrees. Do not touch the gray start button yet. - repatha amgenusa 38

Remove the Repatha prefilled syringe carton from the refrigerator and wait 30 minutes. - repatha amgenusa 42
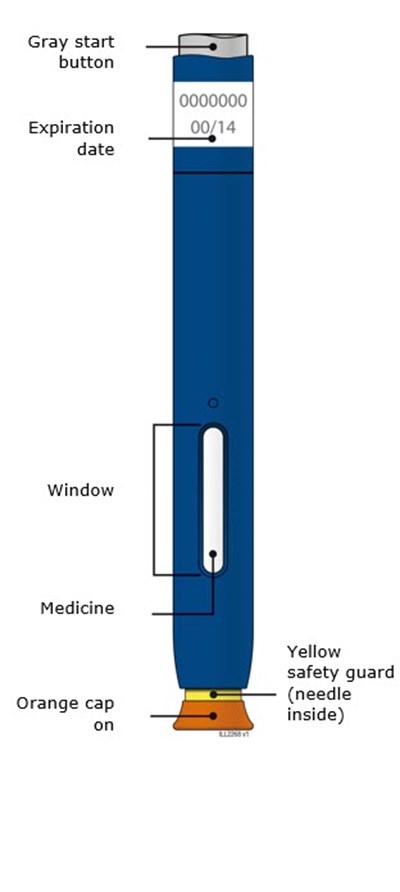
This appears to be a description of a medicine package with a gray button, orange cap, and yellow safety guard. The package also has an expiration date and a window. There is a code resembling "0000000" and "00/14". It is possible that there is a needle inside the package based on the mention of a safety guard. No information about the contents of the medicine or its uses is available.*
1 Rapatha prefilled syringe - repatha amgenusa 43

This appears to be a label of a medical injection which has an expiration date and a yellow window indicating that the injection is complete. There is also a yellow safety guard to protect against the needle, and an orange cap which needs to be taken off.*
Clean your injection site with an alcohol wipe. Let your skin dry before injecting - repatha amgenusa 45

This text is not providing sufficient information to generate a useful description.*
Check the medicine and syringe. - repatha amgenusa 47

This is a list of body parts consisting of Upper arm, Stomach and Thigh.*
PRINCIPAL DISPLAY PANEL1 x 1 mL Prefilled SyringeNDC 72511-750-01Amgen®Repatha® (evolocumab)Injection140 mg/mL140 mg/mLPrefilled SyringeFor Subcutaneous Use OnlyStore at 2°C to 8°C (36°F to 46°F). Do Not Freeze or Shake.Store in Carton to Protect from Light.(see side panel for additional storage information)Sterile Solution – No PreservativeSingle UseRx OnlyCAUTION, See package insert for full prescribing information and Instructions for UseThis Product Contains Dry Natural Rubber.Keep out of the sight and reach of children - repatha amgenusa 50
PRINCIPAL DISPLAY PANEL1 x 3.5 mL Prefilled Cartridge1 On-Body InfusorNDC 72511-770-01Amgen®Repatha® Pushtronex® system(evolocumab)On-Body Infusor and Prefilled Cartridge420 mg/3.5 mL420 mg/3.5 mLFor Subcutaneous Use OnlySingle-Use OnlySterile Solution – No PreservativeStore refrigerated at 2°C to 8°C (36°F to 46°F). Do Not Freeze or Shake.Store in Carton to Protect from Light.(see side panel for additional storage information)Keep out of the sight and reach of children.Refer to Instructions for UseDo Not Use if Package is DamagedOn-Body Infusor Sterilized Using Ethylene OxideKeep DryDo not re-useType BF Applied PartRelative HumidityRange is 15% to 85%Rx Only - repatha amgenusa 51

repatha amgenusa 52
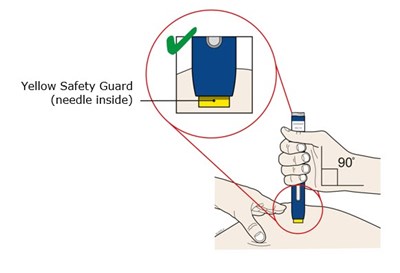
This is a warning label or sign indicating that there is a needle inside the item labeled as "Yellow Safety Guard". It is not clear what type of item this refers to, but it is likely related to healthcare, medical or laboratory equipment that requires caution when handling.*
repatha amgenusa 59
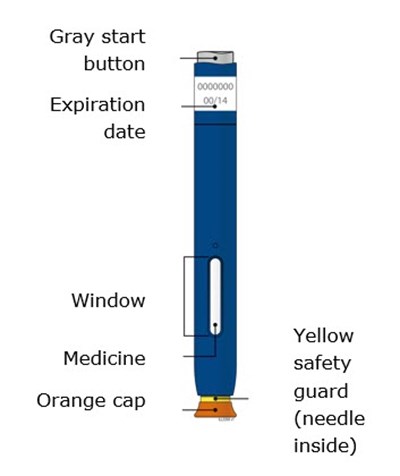
This appears to be a description of a type of medication packaging. The packaging features a gray start button, an expiration date, and a window to view the medicine inside. The medicine may have an orange cap, and there is a yellow safety guard to protect from accidental contact with the included needle.*
repatha amgenusa 60
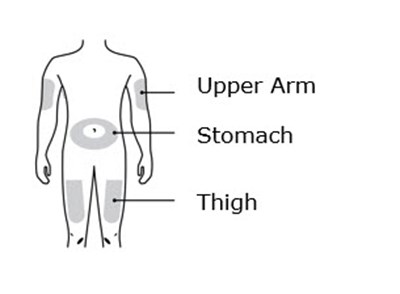
Each word represents a body part, including "Upper Arm", "Stomach", and "Thigh".*
repatha amgenusa 64
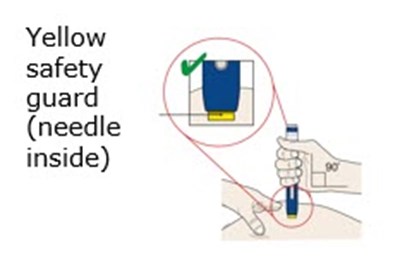
This text appears to describe a yellow safety guard that has a needle inside. It is unclear what this refers to specifically or what its intended use may be.*
repatha amgenusa 69

This is a description of components of a syringe used for administering medicine. The Plunger rod is used to push the medicine out of the Syringe barrel. The Gray needle cap is used to protect the needle. Other details are not available.*
repatha amgenusa 70
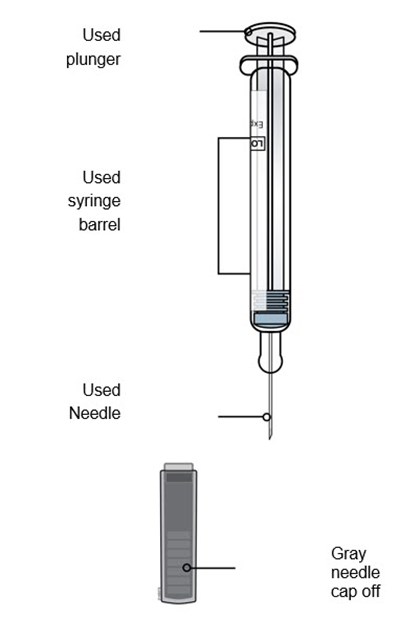
This text is not useful as it contains only a few words that do not form a coherent sentence or description. It seems to mention different items for medical use such as a plunger, syringe barrel, needle, and needle cap.*
repatha amgenusa 84

The text appears to be a mix of different information about a product called Repatha (evolocumab), including dosage, usage instructions, storage guidelines, and cautionary notes about its use. It also mentions that the product comes in prefilled autoinjectors and contains a natural rubber component. However, the output also includes some non-readable characters, making it difficult to provide a comprehensive description.*
repatha amgenusa 85

AMGEN® Repatha (evolocumab) 140 mg/mL, is a sterile solution in a prefilled syringe for subcutaneous use only. The product is stored in a carton to protect it from light and should be kept refrigerated between 2°C to 8°C (6°F to 46°F). The package insert contains full prescribing information and instructions for use. The product contains dry natural rubber, and additional storage information can be found on the side panel. It is essential to keep this product out of the reach of children.*
repatha amgenusa 86

The text seems to be a product description of an Amgen drug called Repatha Pushtronex, which is intended for subcutaneous use only. The package includes a 1 on-body infuser and 1 3.5 mL prefilled cartridge. The drug comes in a sterile solution, without preservatives and should be stored refrigerated. The package also includes additional storage information. However, there are some words and characters that are not readable, and it is not clear what the drug is used for.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.





















































