Product Images Dilaudid
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 11 images provide visual information about the product associated with Dilaudid NDC 76045-009 by Fresenius Kabi Usa, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
dil06 0001 02

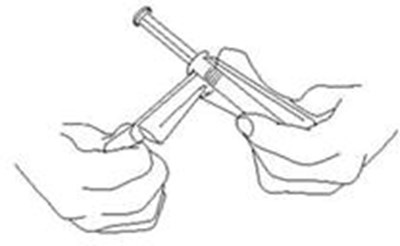
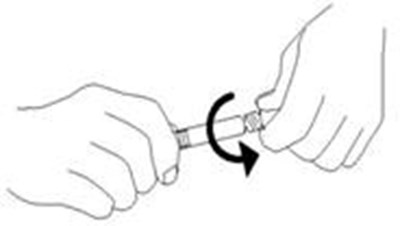
This is a set of instructions for a medical product that contains plastic wrap, a syringe tip cap, and an external collar. The plastic wrap should not be removed, while the syringe tip cap should be removed. The external collar should not be removed.*
dil06 0001 06

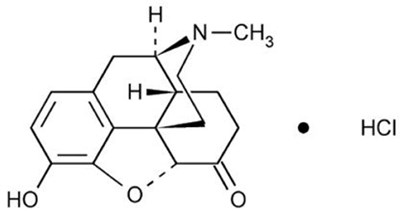
This is a label of Dilaudid® injection which contains 0.5mL of HYDROmorphone HCI. The manufacturer is Prasanius Kabl and the batch number is 403323A.*
dil06 0001 07

This appears to be a label for a single-dose prefilled syringe containing 0.5 grams of injection intended for subcutaneous, intramuscular, and intravenous administration. However, it should not be used for intrathecal or epidural purposes, and it is advised not to place the syringe on a sterile field. The recommended storage temperature is 20°C to 25°C (68°F to 77°F) and should be kept within a controlled room temperature as per USP guidelines.*
dil06 0001 08

The text describes a medication called Dilaudid, which contains Hydrocodone HCl 0.5 mg and can be administered via subcutaneous, intramuscular, or slow intravenous injection. The medication comes in 24 single-dose prefilled syringes, each containing 0.5 mL. It should be noted that the syringe should not be placed on a sterile field and any unused medication should be discarded. The random text "i Rxonly NDC" and "Y peee" appear to be errors and are not useful information.*
dil06 0001 10

This is a description of a medication named Simplist® produced by Fresenius Kabl that comes in a 1L single-dose prefiled syringe. It should be stored at 20° to 25°C and protected from light. The medicine is suitable for intravenous use, however, the unused portion must be discarded, and the syringe should not be placed on a sterile field. There is lot number information available, though some of the characters are unreadable due to .*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.