FDA Label for Fosaprepitant Dimeglumine
View Indications, Usage & Precautions
- 1 INDICATIONS AND USAGE
- 2.1 PREVENTION OF NAUSEA AND VOMITING ASSOCIATED WITH HEC AND MEC IN ADULT PATIENTS
- 2.3 PREPARATION OF FOSAPREPITANT FOR INJECTION
- 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
- 5.1 CLINICALLY SIGNIFICANT CYP3A4 DRUG INTERACTIONS
- 5.2 HYPERSENSITIVITY REACTIONS
- 5.3 INFUSION SITE REACTIONS
- 5.4 DECREASE IN INR WITH CONCOMITANT WARFARIN
- 5.5 RISK OF REDUCED EFFICACY OF HORMONAL CONTRACEPTIVES
- 6 ADVERSE REACTIONS
- 6.1 CLINICAL TRIALS EXPERIENCE
- 6.2 POSTMARKETING EXPERIENCE
- 7.1 EFFECT OF FOSAPREPITANT/APREPITANT ON THE PHARMACOKINETICS OF OTHER DRUGS
- 7.2 EFFECT OF OTHER DRUGS ON THE PHARMACOKINETICS OF FOSAPREPITANT/APREPITANT
- 8.4 PEDIATRIC USE
- 8.5 GERIATRIC USE
- 8.6 PATIENTS WITH HEPATIC IMPAIRMENT
- 10 OVERDOSAGE
- 11 DESCRIPTION
- 12.1 MECHANISM OF ACTION
- 14.1 PREVENTION OF NAUSEA AND VOMITING ASSOCIATED WITH HEC IN ADULTS
- 14.2 PREVENTION OF NAUSEA AND VOMITING ASSOCIATED WITH MEC IN ADULTS
- 16 HOW SUPPLIED/STORAGE AND HANDLING
- 17 PATIENT COUNSELING INFORMATION
- PRINCIPAL DISPLAY PANEL - 150 MG VIAL LABEL
- PRINCIPAL DISPLAY PANEL - 150 MG CARTON LABEL
Fosaprepitant Dimeglumine Product Label
The following document was submitted to the FDA by the labeler of this product Hikma Pharmaceuticals Usa Inc.. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
1 Indications And Usage
Fosaprepitant for Injection, in combination with other antiemetic agents, is indicated in adults for the prevention of:
- acute and delayed nausea and vomiting associated with initial and repeat courses of highly emetogenic cancer chemotherapy (HEC) including high-dose cisplatin.
- delayed nausea and vomiting associated with initial and repeat courses of moderately emetogenic cancer chemotherapy (MEC).
- Fosaprepitant for Injection has not been studied for the treatment of established nausea and vomiting.
Limitations of Use
2.1 Prevention Of Nausea And Vomiting Associated With Hec And Mec In Adult Patients
The recommended dosage of fosaprepitant for injection, dexamethasone, and a 5-HT3 antagonist for the prevention of nausea and vomiting associated with administration of HEC or MEC in adults is shown in Table 1 or Table 2, respectively. Administer fosaprepitant for injection as an intravenous infusion on Day 1 over 20 to 30 minutes, completing the infusion approximately 30 minutes prior to chemotherapy.
| Day 1 | Day 2 | Day 3 | Day 4 | |
|---|---|---|---|---|
| Fosaprepitant for Injection | 150 mg intravenously over 20 to 30 minutes | none | none | none |
| Dexamethasone Administer dexamethasone 30 minutes prior to chemotherapy treatment on Day 1 and in the morning on Days 2 through 4. Also administer dexamethasone in the evenings on Days 3 and 4. A 50% dosage reduction of dexamethasone on Days 1 and 2 is recommended to account for a drug interaction with fosaprepitant for injection [see Clinical Pharmacology (12.3)]. | 12 mg orally | 8 mg orally | 8 mg orally twice daily | 8 mg orally twice daily |
| 5-HT3 antagonist | See selected 5-HT3 antagonist prescribing information for the recommended dosage | none | none | none |
| Day 1 | |
|---|---|
| Fosaprepitant for Injection | 150 mg intravenously over 20 to 30 minutes |
| Dexamethasone Administer dexamethasone 30 minutes prior to chemotherapy treatment on Day 1. A 50% dosage reduction of dexamethasone is recommended to account for a drug interaction with fosaprepitant for injection [see Clinical Pharmacology (12.3)]. | 12 mg orally |
| 5-HT3 antagonist | See selected 5-HT3 antagonist prescribing information for the recommended dosage |
2.3 Preparation Of Fosaprepitant For Injection
| Step 1 | Aseptically inject 5 mL 0.9% Sodium Chloride Injection, USP into the vial. Assure that 0.9% Sodium Chloride Injection, USP is added to the vial along the vial wall in order to prevent foaming. Swirl the vial gently. Avoid shaking and jetting 0.9% Sodium Chloride Injection, USP into the vial. |
| Step 2 | Aseptically prepare an infusion bag filled with 145 mL of 0.9% Sodium Chloride Injection, USP. |
| Step 3 | Aseptically withdraw the entire volume from the vial and transfer it into the infusion bag containing 145 mL of 0.9% Sodium Chloride Injection, USP to yield a total volume of 150 mL and a final concentration of 1 mg/mL. |
| Step 4 | Gently invert the bag 2 to 3 times. |
| Step 5 | Adults The entire volume of the prepared infusion bag (150 mL) should be administered. |
| Step 6 | Before administration, inspect the bag for particulate matter and discoloration. Discard the bag if particulate and/or discoloration are observed. |
Caution: Do not mix or reconstitute fosaprepitant for injection with solutions for which physical and chemical compatibility have not been established. Fosaprepitant for Injection is incompatible with any solutions containing divalent cations (e.g., Ca2+, Mg2+), including Lactated Ringer's Solution and Hartmann's Solution.
3 Dosage Forms And Strengths
Fosaprepitant for Injection: 150 mg fosaprepitant, white to off-white lyophilized powder in single-dose glass vial for reconstitution
4 Contraindications
Fosaprepitant for Injection is contraindicated in patients:
- who are hypersensitive to any component of the product. Hypersensitivity reactions including anaphylactic reactions, flushing, erythema, and dyspnea have been reported [see Warnings and Precautions (5.2), Adverse Reactions (6.2)].
- taking pimozide. Inhibition of CYP3A4 by aprepitant, the active moiety, could result in elevated plasma concentrations of this drug, which is a CYP3A4 substrate, potentially causing serious or life-threatening reactions, such as QT prolongation, a known adverse reaction of pimozide [see Warnings and Precautions (5.1)].
5.1 Clinically Significant Cyp3a4 Drug Interactions
Fosaprepitant, a prodrug of aprepitant, is a weak inhibitor of CYP3A4, and aprepitant is a substrate, inhibitor, and inducer of CYP3A4.
- Use of fosaprepitant for injection with other drugs that are CYP3A4 substrates, may result in increased plasma concentration of the concomitant drug.
- Use of pimozide with fosaprepitant for injection is contraindicated due to the risk of significantly increased plasma concentrations of pimozide, potentially resulting in prolongation of the QT interval, a known adverse reaction of pimozide [see Contraindications (4)].
- Use of fosaprepitant for injection with strong or moderate CYP3A4 inhibitors (e.g., ketoconazole, diltiazem) may increase plasma concentrations of aprepitant and result in an increased risk of adverse reactions related to fosaprepitant for injection.
- Use of fosaprepitant for injection with strong CYP3A4 inducers (e.g., rifampin) may result in a reduction in aprepitant plasma concentrations and decreased efficacy of fosaprepitant for injection.
See Table 7 and Table 8 for a listing of potentially significant drug interactions [see Drug Interactions (7.1, 7.2)].
5.2 Hypersensitivity Reactions
Serious hypersensitivity reactions, including anaphylaxis and anaphylactic shock, during or soon after infusion of fosaprepitant for injection have occurred. Symptoms including flushing, erythema, dyspnea, hypotension and syncope have been reported [see Adverse Reactions (6.2)].
Monitor patients during and after infusion. If hypersensitivity reactions occur, discontinue the infusion and administer appropriate medical therapy. Do not reinitiate fosaprepitant for injection in patients who experience these symptoms with previous use [see Contraindications (4)].
5.3 Infusion Site Reactions
Infusion site reactions (ISRs) have been reported with the use of fosaprepitant for injection [see Adverse Reactions (6.1)]. The majority of severe ISRs, including thrombophlebitis and vasculitis, were reported with concomitant vesicant (anthracycline-based) chemotherapy administration, particularly when associated with extravasation. Necrosis was also reported in some patients with concomitant vesicant chemotherapy. Most ISRs occurred with the first, second or third exposure to single doses of fosaprepitant for injection and in some cases, reactions persisted for two weeks or longer. Treatment of severe ISRs consisted of medical, and in some cases surgical, intervention.
Avoid infusion of fosaprepitant for injection into small veins or through a butterfly catheter. If a severe ISR develops during infusion, discontinue the infusion and administer appropriate medical treatment.
5.4 Decrease In Inr With Concomitant Warfarin
Coadministration of fosaprepitant for injection with warfarin, a CYP2C9 substrate, may result in a clinically significant decrease in the International Normalized Ratio (INR) of prothrombin time [see Clinical Pharmacology (12.3)]. Monitor the INR in patients on chronic warfarin therapy in the 2-week period, particularly at 7 to 10 days, following initiation of fosaprepitant for injection with each chemotherapy cycle [see Drug Interactions (7.1)].
5.5 Risk Of Reduced Efficacy Of Hormonal Contraceptives
Upon coadministration with fosaprepitant for injection, the efficacy of hormonal contraceptives may be reduced during administration of and for 28 days following the last dose of fosaprepitant for injection [see Clinical Pharmacology (12.3)]. Advise patients to use effective alternative or back-up methods of contraception during treatment with fosaprepitant for injection and for 1 month following administration of fosaprepitant for injection [see Drug Interactions (7.1), Use in Specific Populations (8.3)].
6 Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Hypersensitivity Reactions [see Warnings and Precautions (5.2)]
- Infusion Site Reactions [see Warnings and Precautions (5.3)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
The overall safety of fosaprepitant for injection was evaluated in approximately 1600 adult patients.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of fosaprepitant. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Skin and subcutaneous tissue disorders: pruritus, rash, urticaria, Stevens-Johnson syndrome/toxic epidermal necrolysis [see Warnings and Precautions (5.2)].
Immune system disorders: hypersensitivity reactions including anaphylaxis and anaphylactic shock [see Contraindications (4), Warnings and Precautions (5.2)].
Nervous system disorders: ifosfamide-induced neurotoxicity reported after fosaprepitant for injection and ifosfamide coadministration.
7.1 Effect Of Fosaprepitant/Aprepitant On The Pharmacokinetics Of Other Drugs
When administered intravenously, fosaprepitant, a prodrug of aprepitant, is converted to aprepitant within 30 minutes. Therefore, drug interactions following administration of fosaprepitant for injection are likely to occur with drugs that interact with oral aprepitant.
Fosaprepitant, given as a single 150-mg dose, is a weak inhibitor of CYP3A4, and the weak inhibition of CYP3A4 continues for 2 days after single dose administration. Single dose fosaprepitant does not induce CYP3A4. Aprepitant is a substrate, an inhibitor, and an inducer of CYP3A4. Aprepitant is also an inducer of CYP2C9 [see Clinical Pharmacology (12.3)].
Some substrates of CYP3A4 are contraindicated with fosaprepitant for injection [see Contraindications (4)]. Dosage adjustment of some CYP3A4 and CYP2C9 substrates may be warranted, as shown in Table 7.
| CYP3A4 Substrates | |
| Pimozide | |
| Clinical Impact | Increased pimozide exposure |
| Intervention | Fosaprepitant for Injection is contraindicated [see Contraindications (4)]. |
| Benzodiazepines | |
| Clinical Impact | Increased exposure to midazolam or other benzodiazepines metabolized via CYP3A4 (alprazolam, triazolam) may increase the risk of adverse reactions [see Clinical Pharmacology (12.3)]. |
| Intervention | Monitor for benzodiazepine-related adverse reactions. |
| Dexamethasone | |
| Clinical Impact | Increased dexamethasone exposure [see Clinical Pharmacology (12.3)]. |
| Intervention | Reduce the dose of oral dexamethasone by approximately 50% [see Dosage and Administration (2.1)]. |
| Methylprednisolone | |
| Clinical Impact | Increased methylprednisolone exposure [see Clinical Pharmacology (12.3)]. |
| Intervention | Reduce the dose of oral methylprednisolone by approximately 50% on Days 1 and 2 for patients receiving HEC and on Day 1 for patients receiving MEC. Reduce the dose of intravenous methylprednisolone by 25% on Days 1 and 2 for patients receiving HEC and on Day 1 for patients receiving MEC. |
| Chemotherapeutic agents that are metabolized by CYP3A4 | |
| Clinical Impact | Increased exposure of the chemotherapeutic agent may increase the risk of adverse reactions [see Clinical Pharmacology (12.3)]. |
| Intervention | Vinblastine, vincristine, or ifosfamide or other chemotherapeutic agents
|
| Hormonal Contraceptives | |
| Clinical Impact | Decreased hormonal exposure during administration of and for 28 days after administration of the last dose of fosaprepitant for injection [see Warnings and Precautions (5.5), Use in Specific Populations (8.3), and Clinical Pharmacology (12.3)]. |
| Intervention | Effective alternative or back-up methods of contraception (such as condoms and spermicides) should be used during treatment with fosaprepitant for injection and for 1 month following administration of fosaprepitant for injection. |
| Examples | birth control pills, skin patches, implants, and certain IUDs |
| CYP2C9 Substrates | |
| Warfarin | |
| Clinical Impact | Decreased warfarin exposure and decreased prothrombin time (INR) [see Warnings and Precautions (5.4), Clinical Pharmacology (12.3)]. |
| Intervention | In patients on chronic warfarin therapy, monitor the prothrombin time (INR) in the 2-week period, particularly at 7 to 10 days, following administration of fosaprepitant for injection with each chemotherapy cycle. |
| Other | |
| 5-HT3 Antagonists | |
| Clinical Impact | No change in the exposure of the 5-HT3 antagonist [see Clinical Pharmacology (12.3)]. |
| Intervention | No dosage adjustment needed |
| Examples | ondansetron, granisetron, dolasetron |
7.2 Effect Of Other Drugs On The Pharmacokinetics Of Fosaprepitant/Aprepitant
Aprepitant is a CYP3A4 substrate [see Clinical Pharmacology (12.3)]. Co-administration of fosaprepitant for injection with drugs that are inhibitors or inducers of CYP3A4 may result in increased or decreased plasma concentrations of aprepitant, respectively, as shown in Table 8.
| Moderate to Strong CYP3A4 Inhibitors | |
| Clinical Impact | Significantly increased exposure of aprepitant may increase the risk of adverse reactions associated with fosaprepitant for injection [see Adverse Reactions (6.1), Clinical Pharmacology (12.3)]. |
| Intervention | Avoid concomitant use of fosaprepitant for injection |
| Examples | Moderate inhibitor: diltiazem Strong inhibitors: ketoconazole, itraconazole, nefazodone, troleandomycin, clarithromycin, ritonavir, nelfinavir |
| Strong CYP3A4 Inducers | |
| Clinical Impact | Substantially decreased exposure of aprepitant in patients chronically taking a strong CYP3A4 inducer may decrease the efficacy of fosaprepitant for injection [see Clinical Pharmacology (12.3)]. |
| Intervention | Avoid concomitant use of fosaprepitant for injection |
| Examples | rifampin, carbamazepine, phenytoin |
8.4 Pediatric Use
The safety and effectiveness of fosaprepitant for injection for the prevention of nausea and vomiting associated with HEC or MEC have not been established in patients less than 6 months of age.
Juvenile Animal Toxicity Data
In juvenile dogs treated with fosaprepitant, changes in reproductive organs were observed. In juvenile rats treated with aprepitant, slight changes in sexual maturation were observed without an effect on reproduction. No effects on neurobehavior, sensory and motor function, or learning and memory were observed in rats.
In a toxicity study in juvenile dogs treated with fosaprepitant from postnatal day 14 (equivalent to a newborn human) to day 42 (approximately equivalent to a 2 year old human), decreased testicular weight and Leydig cell size were seen in the males at 6 mg/kg/day and increased uterine weight, hypertrophy of the uterus and cervix, and edema of vaginal tissues were seen in females from 4 mg/kg/day. A study was also conducted in young rats to evaluate the effects of aprepitant on growth and on neurobehavioral and sexual development. Rats were treated at oral doses up to the maximum feasible dose of 1000 mg/kg twice daily from the early postnatal period (Postnatal Day 10 (equivalent to a newborn human) through Postnatal Day 58 (approximately equivalent to a 15 year old human)). Slight changes in the onset of sexual maturation were observed in female and male rats; however, there were no effects on mating, fertility, embryonic-fetal survival, or histomorphology of the reproductive organs. There were no effects in neurobehavioral tests of sensory function, motor function, and learning and memory.
Pediatric use information is approved for Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc.’s Emend (fosaprepitant) for injection. However, due to Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc.’s marketing exclusivity rights, this drug product is not labeled with that pediatric information.
8.5 Geriatric Use
Of the 1649 adult cancer patients treated with intravenous fosaprepitant for injection in HEC and MEC clinical studies, 27% were aged 65 and over, while 5% were aged 75 and over. Other reported clinical experience with fosaprepitant for injection has not identified differences in responses between elderly and younger patients. In general, use caution when dosing elderly patients as they have a greater frequency of decreased hepatic, renal or cardiac function and concomitant disease or other drug therapy [see Clinical Pharmacology (12.3)].
8.6 Patients With Hepatic Impairment
The pharmacokinetics of aprepitant in patients with mild and moderate hepatic impairment were similar to those of healthy subjects with normal hepatic function. No dosage adjustment is necessary for patients with mild to moderate hepatic impairment (Child-Pugh score 5 to 9). There are no clinical or pharmacokinetic data in patients with severe hepatic impairment (Child-Pugh score greater than 9). Therefore, additional monitoring for adverse reactions in these patients may be warranted when fosaprepitant for injection is administered [see Clinical Pharmacology (12.3)].
10 Overdosage
There is no specific information on the treatment of overdosage with fosaprepitant or aprepitant.
In the event of overdose, fosaprepitant for injection should be discontinued and general supportive treatment and monitoring should be provided. Because of the antiemetic activity of fosaprepitant for injection, drug-induced emesis may not be effective in cases of fosaprepitant for injection overdosage.
Aprepitant is not removed by hemodialysis.
11 Description
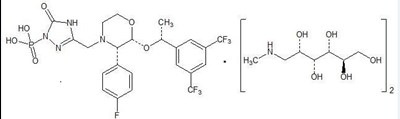
Fosaprepitant for Injection is a sterile, lyophilized formulation containing fosaprepitant dimeglumine, a prodrug of aprepitant, a substance P/neurokinin-1 (NK1) receptor antagonist, an antiemetic agent, chemically described as 1-Deoxy-1-(methylamino)-D-glucitol[3-[[(2R,3S)-2-[(1R)-1-[3,5-bis(trifluoromethyl)phenyl]ethoxy]-3-(4-fluorophenyl)-4-morpholinyl]methyl]-2,5-dihydro-5-oxo-1H-1,2,4-triazol-1-yl]phosphonate (2:1) (salt).
Its empirical formula is C23H22F7N4O6P ∙ 2(C7H17NO5) and its structural formula is:
Fosaprepitant dimeglumine is a white or off-white amorphous powder with a molecular weight of 1004.83. It is freely soluble in water.
Each vial of fosaprepitant for injection for administration as an intravenous infusion contains 150 mg of fosaprepitant (equivalent to 245.3 mg of fosaprepitant dimeglumine) and the following inactive ingredients: edetate disodium (5.4 mg), polysorbate 80 (75 mg), lactose anhydrous (375 mg), sodium hydroxide and/or hydrochloric acid (for pH adjustment).
12.1 Mechanism Of Action
Fosaprepitant is a prodrug of aprepitant and accordingly, its antiemetic effects are attributable to aprepitant.
Aprepitant is a selective high-affinity antagonist of human substance P/neurokinin 1 (NK1) receptors. Aprepitant has little or no affinity for serotonin (5-HT3), dopamine, and corticosteroid receptors, the targets of existing therapies for chemotherapy-induced nausea and vomiting (CINV). Aprepitant has been shown in animal models to inhibit emesis induced by cytotoxic chemotherapeutic agents, such as cisplatin, via central actions. Animal and human Positron Emission Tomography (PET) studies with aprepitant have shown that it crosses the blood brain barrier and occupies brain NK1 receptors. Animal and human studies have shown that aprepitant augments the antiemetic activity of the 5-HT3-receptor antagonist ondansetron and the corticosteroid dexamethasone and inhibits both the acute and delayed phases of cisplatin-induced emesis.
14.1 Prevention Of Nausea And Vomiting Associated With Hec In Adults
In a randomized, parallel, double-blind, active-controlled study, fosaprepitant for injection 150 mg as a single intravenous infusion (N=1147) was compared to a 3-day oral aprepitant regimen (N=1175) in patients receiving a HEC regimen that included cisplatin (≥70 mg/m2). All patients in both groups received dexamethasone and ondansetron (see Table 11). Patient demographics were similar between the two treatment groups. Of the total 2322 patients, 63% were men, 56% White, 26% Asian, 3% American Indian/Alaska Native, 2% Black, 13% Multi-Racial, and 33% Hispanic/Latino ethnicity. Patient ages ranged from 19 to 86 years of age, with a mean age of 56 years. Other concomitant chemotherapy agents commonly administered were fluorouracil (17%), gemcitabine (16%), paclitaxel (15%), and etoposide (12%).
| Day 1 | Day 2 | Day 3 | Day 4 | |
|---|---|---|---|---|
| Fosaprepitant for Injection Regimen | ||||
| Fosaprepitant for Injection | 150 mg intravenously over 20 to 30 minutes approximately 30 minutes prior to chemotherapy | none | none | none |
| Oral dexamethasone
Dexamethasone was administered 30 minutes prior to chemotherapy treatment on Day 1 and in the morning on Days 2 through 4. Dexamethasone was also administered in the evenings on Days 3 and 4. The 12 mg dose of dexamethasone on Day 1 and the 8 mg once daily dose on Day 2 reflects a dosage adjustment to account for a drug interaction with the fosaprepitant for injection regimen [see Clinical Pharmacology (12.3)]. | 12 mg | 8 mg | 8 mg twice daily | 8 mg twice daily |
| Ondansetron | Ondansetron Ondansetron 32 mg intravenous was used in the clinical trials of fosaprepitant. Although this dose was used in clinical trials, this is no longer the currently recommended dose. Refer to the ondansetron prescribing information for the current recommended dose. | none | none | none |
| Oral Aprepitant Regimen | ||||
| Aprepitant capsules | 125 mg | 80 mg | 80 mg | none |
| Oral dexamethasone
Dexamethasone was administered 30 minutes prior to chemotherapy treatment on Day 1 and in the morning on Days 2 through 4. The 12 mg dose of dexamethasone on Day 1 and the 8 mg once daily dose on Days 2 through 4 reflects a dosage adjustment to account for a drug interaction with the oral aprepitant regimen [see Clinical Pharmacology (12.3)]. | 12 mg | 8 mg | 8 mg | 8 mg |
| Ondansetron | Ondansetron | none | none | none |
The efficacy of fosaprepitant for injection was evaluated based on the primary and secondary endpoints listed in Table 12 and was shown to be non-inferior to that of the 3-day oral aprepitant regimen with regard to complete response in each of the evaluated phases. The pre-specified non-inferiority margin for complete response in the overall phase was 7%. The pre-specified non-inferiority margin for complete response in the delayed phase was 7.3%. The pre-specified non-inferiority margin for no vomiting in the overall phase was 8.2%.
| ENDPOINTS | Fosaprepitant for Injection Regimen (N = 1106) N: Number of patients included in the primary analysis of complete response. % | Oral Aprepitant Regimen (N = 1134) % | Difference Difference and Confidence interval (CI) were calculated using the method proposed by Miettinen and Nurminen and adjusted for Gender. (95% CI) |
|---|---|---|---|
| PRIMARY ENDPOINT | |||
| Complete Response Complete Response = no vomiting and no use of rescue therapy. | |||
| Overall Overall = 0 to 120 hours post-initiation of cisplatin chemotherapy. | 71.9 | 72.3 | -0.4 (-4.1, 3.3) |
| SECONDARY ENDPOINTS | |||
| Complete Response | |||
| Delayed phase Delayed phase = 25 to 120 hours post-initiation of cisplatin chemotherapy. | 74.3 | 74.2 | 0.1 (-3.5, 3.7) |
| No Vomiting | |||
| Overall | 72.9 | 74.6 | -1.7 (-5.3, 2.0) |
14.2 Prevention Of Nausea And Vomiting Associated With Mec In Adults
In a randomized, parallel, double-blind, active comparator-controlled study, fosaprepitant for injection 150 mg as a single intravenous infusion (N=502) in combination with ondansetron and dexamethasone (fosaprepitant for injection regimen) was compared with ondansetron and dexamethasone alone (standard therapy) (N=498) (see Table 13) in patients receiving a MEC regimen. Patient demographics were similar between the two treatment groups. Of the total 1,000 patients included in the efficacy analysis, 41% were men, 84% White, 4% Asian, 1% American Indian/Alaska Native, 2% Black, 10% Multi-Racial, and 19% Hispanic/Latino ethnicity. Patient ages ranged from 23 to 88 years of age, with a mean age of 60 years. The most commonly administered MEC chemotherapeutic agents were carboplatin (51%), oxaliplatin (24%), and cyclophosphamide (12%).
| Day 1 | Day 2 | Day 3 | |
|---|---|---|---|
| Fosaprepitant for Injection Regimen | |||
| Fosaprepitant for Injection | 150 mg intravenously over 20 to 30 minutes approximately 30 minutes prior to chemotherapy | none | none |
| Oral Dexamethasone Dexamethasone was administered 30 minutes prior to chemotherapy treatment on Day 1. The 12 mg dose reflects a dosage adjustment to account for a drug interaction with the fosaprepitant for injection regimen [see Clinical Pharmacology (12.3)]. | 12 mg | none | none |
| Oral Ondansetron The first ondansetron dose was administered 30 to 60 minutes prior to chemotherapy treatment on Day 1 and the second dose was administered 8 hours after first ondansetron dose. | 8 mg for 2 doses | none | none |
| Standard Therapy | |||
| Oral Dexamethasone | 20 mg | none | none |
| Oral Ondansetron | 8 mg for 2 doses | 8 mg twice daily | 8 mg twice daily |
The primary endpoint was complete response (defined as no vomiting and no rescue therapy) in the delayed phase (25 to 120 hours) of chemotherapy-induced nausea and vomiting. The results by treatment group are shown in Table 14.
| ENDPOINTS | Fosaprepitant for Injection Regimen (N = 502) N: Number of patients included in the intention to treat population. % | Standard Therapy Regimen (N = 498) % | P-Value | Treatment Difference (95% CI) |
|---|---|---|---|---|
| PRIMARY ENDPOINT | ||||
| Complete Response Complete Response = no vomiting and no use of rescue therapy. | ||||
| Delayed phase Delayed phase = 25 to 120 hours post-initiation of chemotherapy. | 78.9 | 68.5 | <0.001 | 10.4 (5.1, 15.9) |
16 How Supplied/Storage And Handling
Single-dose glass vial containing 150 mg of fosaprepitant as a white to off-white lyophilized powder for reconstitution. Supplied as follows:
| NDC 0143-9384-01 | 1 vial per carton. |
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Principal Display Panel - 150 Mg Vial Label
NDC 0143-9384-01 Rx only
Fosaprepitant
for Injection
150 mg per vial
Single Dose Vial
Discard unused portion
Principal Display Panel - 150 Mg Carton Label
NDC 0143-9384-01 Rx only
Fosaprepitant
for Injection
150 mg per vial
Reconstitute with 5 mL 0.9%
Sodium Chloride for Injection
STERILE LYOPHILIZED POWDER
FOR INTRAVENOUS USE ONLY AFTER
RECONSTITUTION AND DILUTION
DO NOT USE WITH SOLUTIONS
CONTAINING DIVALENT CATIONS
(e.g., Ca2+, Mg2+) INCLUDING
LACTATED RINGER'S SOLUTION
AND HARTMANN'S SOLUTION.
Single Dose Vial
Discard unused portion
* Please review the disclaimer below.