Product Images Vonvendi
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 38 images provide visual information about the product associated with Vonvendi NDC 0944-7553 by Takeda Pharmaceuticals America, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
vonvendi 26

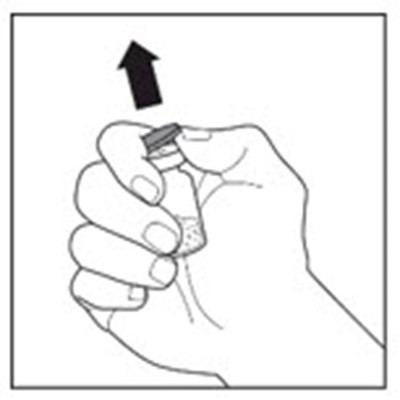
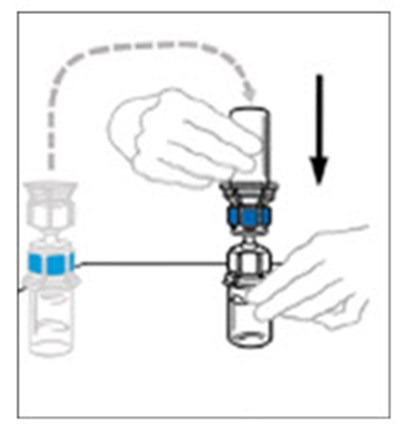
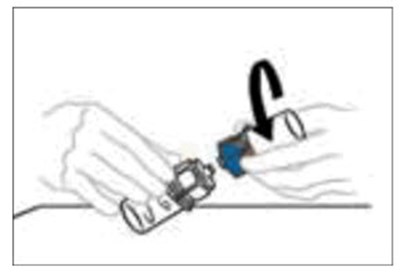
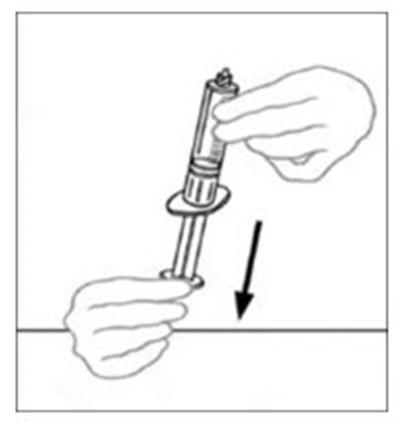
The text appears to describe a "syringe tip", which is a small, pointed end of a syringe that is used for injecting or dispensing liquids. The copyright symbol suggests that this may be a proprietary term or design belonging to a particular company or manufacturer.*
vonvendi 33

VONVENDI is a recombinant von Willebrand factor product by BaxaltaUs Inc. The product is non-sterile and non-pyrogenic. The actual potency of the product is not specified in the text. The product is a freeze-dried powder and requires reconstitution before use. The text includes a lot number (G855-65-008-1) and an expiration date.*
vonvendi 34

VONVENDI is a single-use lyophilized powder of von Willebrand factor (Recombinant) for intravenous administration after reconstitution only. It is manufactured by Baxalta US Inc. and has an NDC code of 0944-7550-01. The label also contains information on lot number and expiration date.*
vonvendi 35

This is a description of a product that appears to be sterile water for injection. The product code is nbc09s4a-0013-05, and it is intended for use in reconstituting another product. The container is a single-dose and nonpyrogenic, but no antimicrobial agent or other substance appears to have been added to the water. The product should not be used for intravascular injection without the addition of a suitable solute. There is also a lot number and expiration date provided.*
vonvendi 36

VONVENDI is a recombinant treatment for von Willebrand factor (VWF) deficiency, a bleeding disorder. The product is stored at room temperature and should not be frozen. One package contains 1 g of the medicine, and its components do not contain natural rubber latex. It is not available if further details are missing.*
vonvendi 37

VONVENDI is a recombinant von Willebrand factor used for intravenous administration after reconstitution. It is sterile, in single-use vials and comes as a lyophilized powder. The lot number is NO COAT °§ H. Its NDC is 0944-7552-01 or 0344755201. VONVENDI is manufactured by Baxalta US Inc. located in Lexington, MA.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.