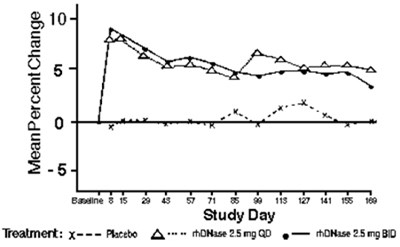
Product Images Pulmozyme
View Photos of Packaging, Labels & Appearance
- Figure 1 - pulmozyme 01
- Figure A - pulmozyme 02
- Figure B - pulmozyme 03
- Figure C - pulmozyme 04
- Figure D - pulmozyme 05
- Figure E - pulmozyme 06
- Figure F - pulmozyme 07
- Figure G - pulmozyme 08
- Figure H - pulmozyme 09
- Figure I - pulmozyme 10
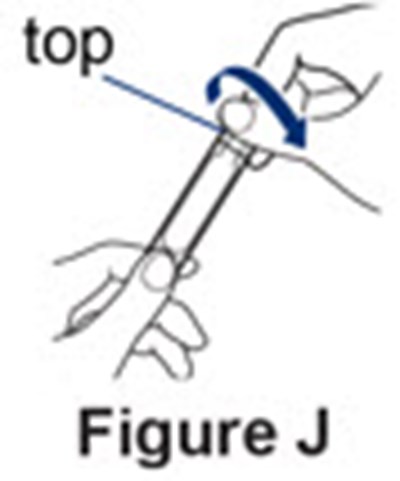
- Figure J - pulmozyme 11
- Figure K - pulmozyme 12
- Figure L - pulmozyme 13
- Figure M - pulmozyme 14
- Figure N - pulmozyme 15
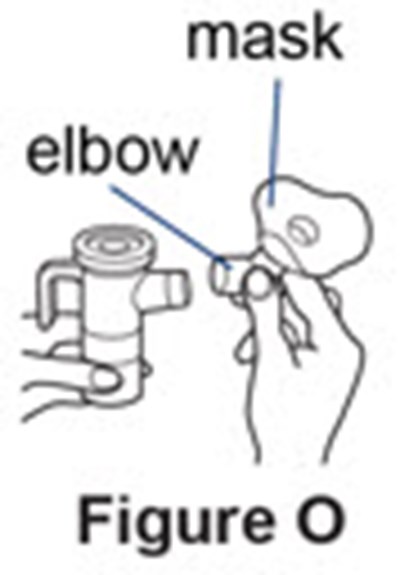
- Figure O - pulmozyme 16
- Figure P - pulmozyme 17
- Figures Q - pulmozyme 18
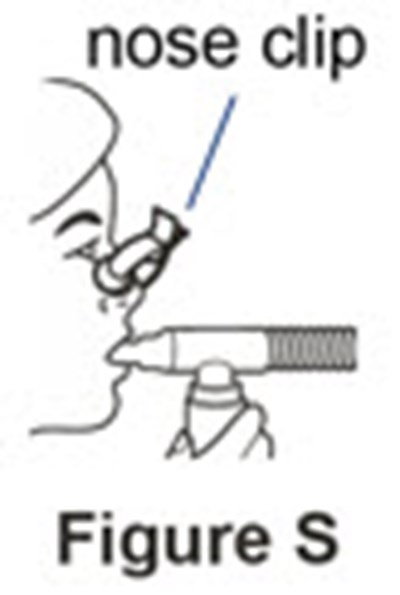
- Figure S - pulmozyme 20
- Figure T - pulmozyme 21
- Figure U - pulmozyme 22
- Figure V - pulmozyme 23
- Figure W - pulmozyme 24
- Figure X - pulmozyme 25
- Figure Y - pulmozyme 26
- Figure A - pulmozyme 27
- Figure B - pulmozyme 28
- Figure C - pulmozyme 29
- Figure D - pulmozyme 30
- Figure E - pulmozyme 31
- Figure F - pulmozyme 32
- Figure G - pulmozyme 33
- Figure H - pulmozyme 34
- Figure I - pulmozyme 35
- Figure J - pulmozyme 36
- Figure K - pulmozyme 37
- Figure L - pulmozyme 38
- Figure M - pulmozyme 39
- Figure N - pulmozyme 40
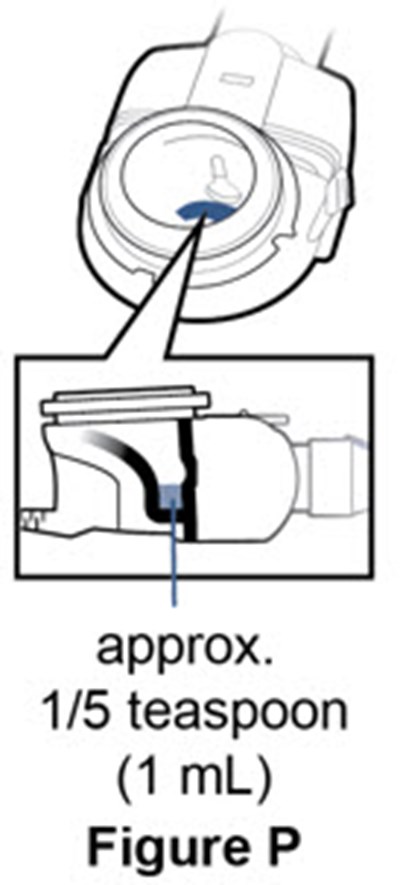
- Figure O - pulmozyme 41
- Figure P - pulmozyme 42
- Figure Q - pulmozyme 43
- PRINCIPAL DISPLAY PANEL - 2.5 mL Ampule Pouch Carton - pulmozyme 44
Product Label Images
The following 43 images provide visual information about the product associated with Pulmozyme NDC 50242-100 by Genentech, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure D - pulmozyme 05
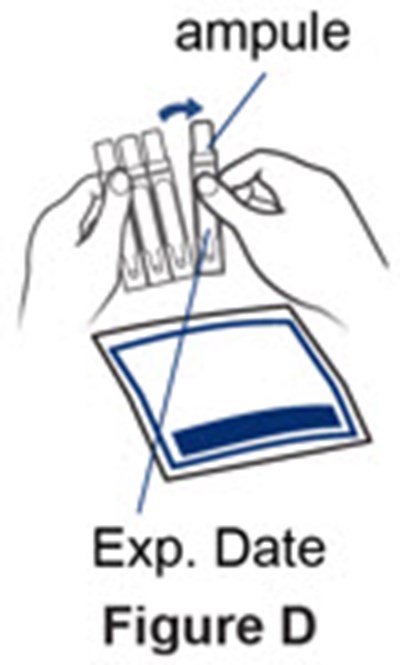
The text is describing a possible product called "ampule" and providing information about its expiration date. There is also a reference to "Figure D", but without additional information, it is not possible to determine its meaning or content.*
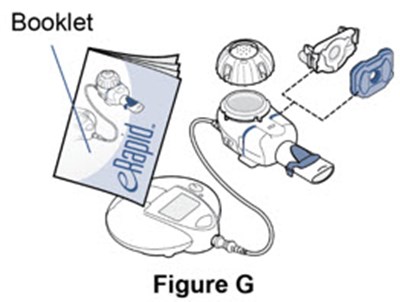
Figure G - pulmozyme 08
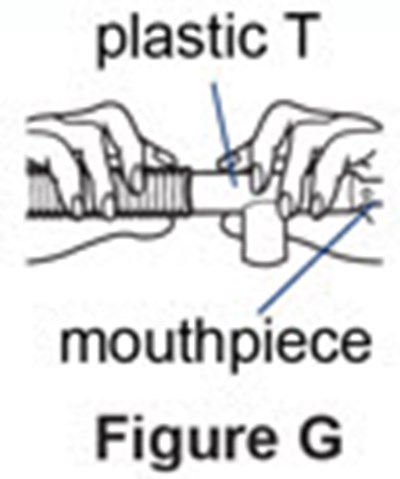
This text is not sufficient to generate a useful description as it contains only a few words and lacks context.*
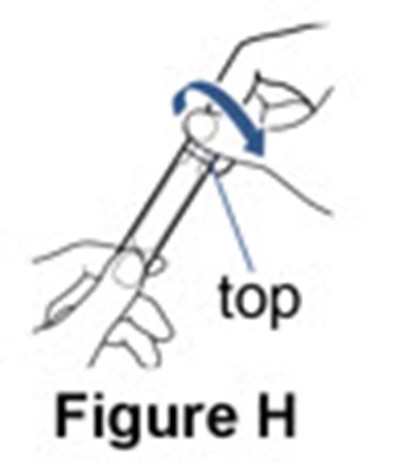
Figure H - pulmozyme 09

This is a simple description that mentions a "nebulizer cup" and a "Figure H". However, without additional context or information, it is unclear what the nebulizer cup does or what Figure H represents.*
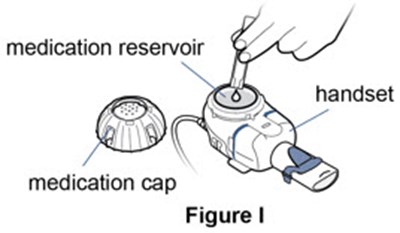
Figure I - pulmozyme 10
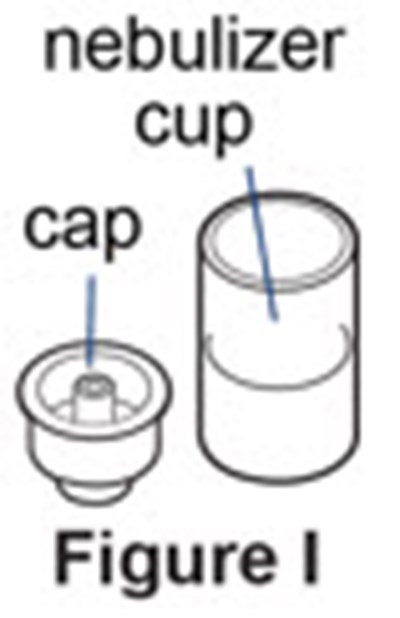
This is a description of a nebulizer cup. There is an image labelled "Figure 1" and the text includes the words "nebulizer" and "cup". However, the text "&0" is not clear and could not be used to provide further information on the description.*
Figure M - pulmozyme 14
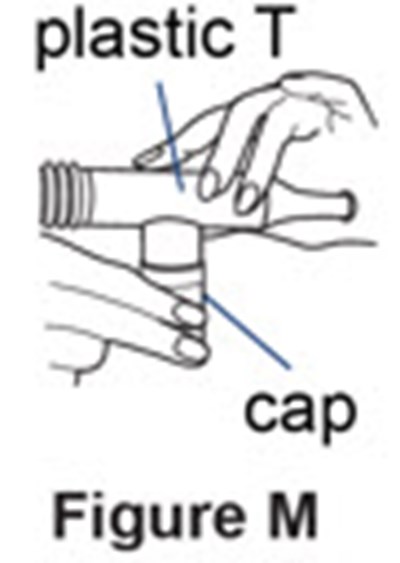
This text indicates that there is a plastic cap with the letter "T" present in a figure labeled "M". However, without additional context, it is difficult to determine the purpose or location of this item.*
Figure N - pulmozyme 15

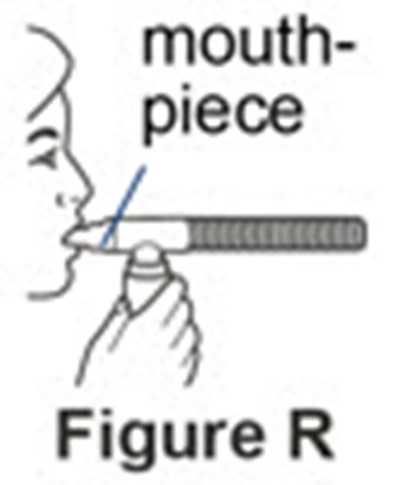
The text describes a "mouthpiece" and features a "figure N". No further information is available.*
Figure V - pulmozyme 23
It appears that this text is not readable and therefore the description cannot be generated.*
Figure A - pulmozyme 27

This text appears to be a list of components related to a medical device, specifically a nebulizer system called "eRapid". The components include a Pulmozyme ampule, instruction booklet, nose clip, medication reservoir and cap, handset, button, light, and an AC power supply. It seems that the nebulizer system can be powered both by batteries and by plugging in the AC power supply. There is also a diagram, labeled as Figure A, possibly illustrating the different components of the nebulizer system.*
Figure C - pulmozyme 29

The text describes an illustration, labeled as "Figure C", showing an outlet and a power supply. No further information is available.*
Figure N - pulmozyme 40

This is a product called "nose clip". There is no further information available.*
PRINCIPAL DISPLAY PANEL - 2.5 mL Ampule Pouch Carton - pulmozyme 44

This is a description of a pharmaceutical product called Pulmozyme Inhalation Solution with the active ingredient Dornase Alfa. The NDC code for this product is 50242-100-40, and it is manufactured by Genentech Inc. The product should be stored in a refrigerated state and was made in Singapore. The text also includes a product identifier code of 10242745 and a US license number of 1048.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.