Product Images Caldolor
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 6 images provide visual information about the product associated with Caldolor NDC 66220-287 by Cumberland Pharmaceuticals Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
cal05 0011 03

Caldolor' is a medication designed for intravenous use, which contains ibuprofen. It comes in a single dose vial, and any unused portion should be discarded. It is essential to dilute it before use. The package insert should be consulted for dosage, dilution, and administration information. It should be stored at a controlled room temperature of 20°C-25°C (68°F-77°F). The lot number, expiration date, and other relevant information are printed on the label. The medication is manufactured for a company called Cumberland Pharmaceuticals based in Nashville, Tennessee, USA.*
cal05 0011 04
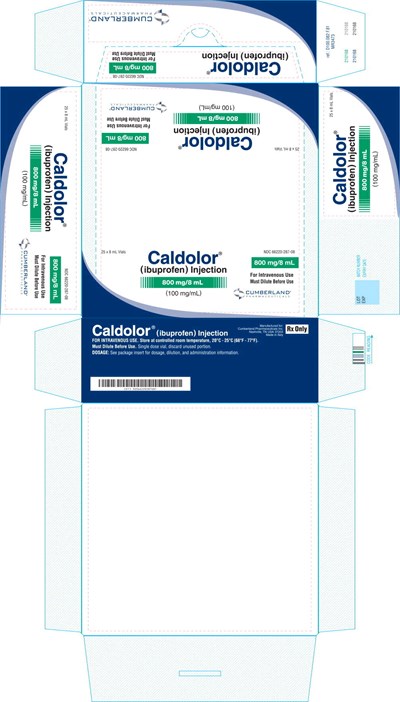
This is a description of Caldolor LR, an injection of Ibuprofen used for intravenous use. The recommended storage temperature is between 20°C-25°C. The dosage information is not available. The text suggests that the injection needs to be diluted before use.*
cal05 0011 05

This text describes details about CALDOLOR(ibuprofen) Injection, which is available in 200mL single dose containers with 800mg/200mL concentration. The medication is for intravenous use only and the package insert should be referred for dosage information. A clear solution should be used and any unused portion should be discarded. It should be stored at a controlled temperature between 20°C-25°C (68°F-77°F). This medication is manufactured for CUMBERLAND® PHARMACEUTICALS in Nashville, TN, USA. No dosage or usage instructions were provided in the text.*
cal05 0011 06

CALDOLOR is a prescription medication that contains 800mg of ibuprofen in each 200mL bag for injection. It is manufactured by Cumberland Pharmaceuticals in Nashville, TN and produced in Spain. This injection solution has an isosmotic pH 7.4 phosphate-buffered sterile solution. CALDOLOR is supplied in single-dose containers and is intended for immediate use. This medication is used for pain relief and is only available by a physician's prescription. Please refer to the package insert for proper dosing information, and store it in a controlled room temperature environment. Use the prescribed dosage and discard the unused portion. The information on LOT and EXP numbers and the NDC code are provided on the package.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.

